Credit: Bionic Vision System
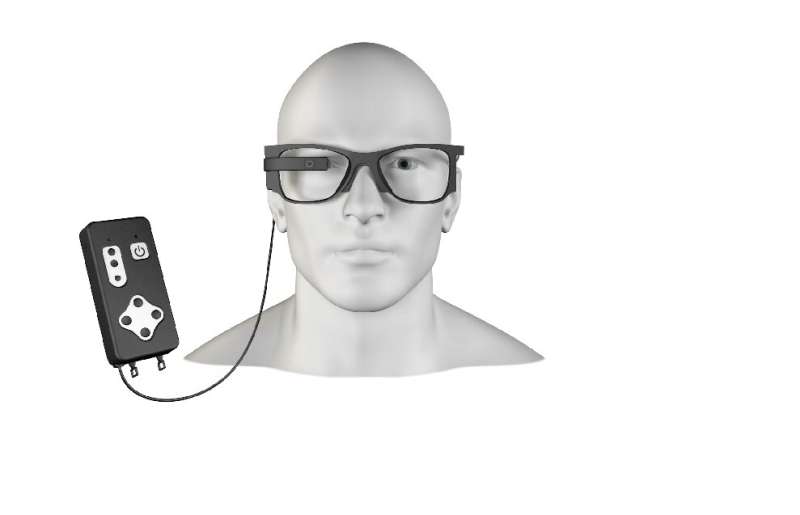
A company focused on a bionic vision system sees hope for improvements in patients by means of a wireless sub-retinal implant together with augmented reality glasses.
Pixium Vision has developed the Prima system in response to age-related macular degeneration. Their system "aims to elicit useful prosthetic bionic vision by electrical stimulation of the retina and therefore partially compensating the loss of natural central vision," they stated.
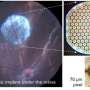
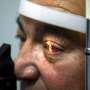
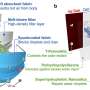
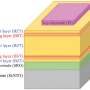
Founded in 2011, the France-based company's system is all about a miniaturized wireless photovoltaic sub-retinal implant. Photovoltaic sub-retinal implant? What does that mean? This involves the conversion of light into electricity; the chip has 378 electrodes implanted under the retina, directly where the photoreceptors had degenerated.
Macular degeneration slowly eats away at the central vision and the person is left with just a blurry or dark hole in the middle of the field of view. Their bionic vision system in clinical testing, targeting dry-age macular degeneration, involves a minimally invasive surgical procedure.
CEO Lloyd Diamond further discussed their technique.
"By implanting the chip directly at the level of photoreceptors [sub-retinal implantation], the subretinal stimulations target the bipolar cells first, before signals reach the gaglion cells; therefore, we are aiming for a more precise and physiological signal processing."
The other key component of their system: augmented reality glasses.
Pixium's team described the interplay between the glasses and chip. "The 2x2 millimeter wide, 30-micron thick photovoltaic chip contains 378 electrodes. Implanted under the retina...it acts like an array of a tiny solar panel powered by pulsed near infrared light projected from a miniature projector transmitting images captured on a mini camera. The camera and projector technologies are integrated into augmented reality glasses."
The photovoltaic cells of the chip converts this optical information into electrical stimulation to 'excite the bipolar nerve cells of the inner retina' and subsequently generates visual perception in the brain," said NS Medical Devices
A clinical trial of their system in France involved five atrophic age-related macular degeneration patients. Admitting criteria included patients 60 years or older, visual acuity of 20/400 or worse and no foveal perception.
Results have come in from the first feasibility trial of Prima System in age-related macular degeneration patients: Most of the patients could identify letters and some sequences of letters. There were no adverse events.
The company wrote in a news release datelined Paris, July 18: "None of the patients had remaining central visual activity at enrolment. Patients had visual rehabilitation, and, at 12-months, most can identify letters with some able to identify sequence of letters and demonstrating increasing speed over time. There were no device-related serious adverse events."
CEO Lloyd Diamond was encouraged by the fact that patients were able to re-establish light perception and also identify letters.
The image created by the implant is just 378 pixels in total. Luke Dormehl's assessment in Digital Trends: This was far from an outright restoration of restoring of 20/20 vision, but it did represent a significant improvement.
The company CEO said "most are seeing light and even patterns, numbers, or letters where they didn't before."
All in all, the company reported that "The 12-month data, after implantation and rehabilitation for five patients in France with advanced dry AMD (geographic atrophy), demonstrated that the PRIMA System met the primary endpoint which showed the successful elicitation of light perception in the central retinal area in all subjects measured by the Octopus test."
CEO Diamond said that "we are proceeding to test feasibility for new upgrades to the system in parallel in both Europe and the US. We will use these new clinical feasibility tests of the upgraded Prima System as the basis for a pivotal study starting in H1 2020."
Diamond and team clearly are pursuing their clinical program; a larger scale study is planned for 2020 in Europe; the goal is a market launch in Europe and the US in the mid-term, said NS Medical Devices.
In the company business update of the July 25 news release, the company said "Pixium Vision will undertake feasibility testing of the upgraded Prima System in the five patients enrolled in the French study and amend feasibility study in a similar set of five patients in the US. Data from the studies will allow the Company to initiate the European pivotal study, which is now expected to start in H1 2020."
© 2019 Science X Network