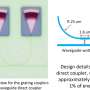
Still from a video showing electrodewetting. Credit: University of California, Los Angeles
When medical laboratories analyze blood samples for signs of disease, they sometimes use instruments that rely on a technology called digital microfluidics. The technique uses electric signals to pull tiny droplets of the sample across a surface so they can be analyzed.
One drawback of the process is that the electric signals tend to damage the surface that the droplets travel over, which can cause the device to fail unexpectedly or deteriorate over time.
Now, a research team led by UCLA engineers and scientists has demonstrated that digital microfluidic devices could be made much more durable if they use the electric signals to push, instead of pull, the droplets over the surface.
A paper about the work was published in Nature. The advance could lead to more powerful and reliable analytical tools for biochemical laboratories and environmental monitoring. It could also improve the durability of the liquid-based lenses used in devices like barcode readers and dental cameras. Those lenses can quickly adjust their focus because they contain a clear droplet that quickly adjusts its shape in response to electrical signals.
Digital microfluidic technology has been studied for nearly two decades; it first appeared in commercial lenses about 10 years ago and in diagnostic instruments more recently. To date, the devices have used water-repelling, or hydrophobic, surfaces, which cause water to bead up—similar to how it behaves on a nonstick cooking pan.
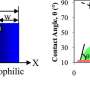
On hydrophobic surfaces, applying electric voltage to one end of a beaded droplet causes that end to be attracted to the surface and flatten out—a phenomenon called electrowetting. That occurs because water can conduct electricity, and a droplet is small enough that its surface tension keeps it together as one unit. That, combined with the fact that the other end of the droplet is still repelled by the surface, causes the entire droplet to move toward the flattened end, in effect "pulling" the liquid toward where electric voltage is applied.
However, most materials are hydrophilic—when water droplets are placed on them, they naturally flatten out—so digital microfluidic devices use surfaces that are coated with a thin hydrophobic layer. But those coatings are prone to fail because voltage can cause them to degrade or crack.
To solve that problem, researchers led by Chang-Jin "CJ" Kim, the Volgenau Professor of Engineering at the UCLA Samueli School of Engineering, set out to make droplets move on a surface without a hydrophobic coating.
Credit: University of California, Los Angeles
"If one could push a liquid droplet from its rear, rather than pulling it from its front, the surface would not need to be hydrophobic," he said.
The problem, Kim said, was that an electric signal can only be used to pull a droplet toward where the voltage is applied—it can't be used to push a droplet away.
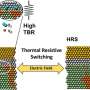
The researchers' solution was to add a tiny amount of electrically charged surfactant to the liquid. (A surfactant is a substance whose molecules repel water on one end and attracts it on the other—soap is an example.) Using the charge, the engineers could use electrical signals to move the surfactant within the droplet.
"Using only electric signals, we can attract the surfactant molecules inside the droplet onto a hydrophilic surface to convert any portions of it to be hydrophobic," said Kim, who also is a member of the California NanoSystems Institute at UCLA.
They called the process electrodewetting—a name they chose to emphasize that it's the opposite of the standard electrowetting technique.
Applying voltage to one end of the droplet on a hydrophilic surface caused the charged surfactant molecules to crowd there, which in turn pushed the droplet up from the surface and then forward, away from where the electricity is applied. That, in effect, made the droplet able to bead on one end, and move across the surface without the need for a special topcoat.
By reversing the direction of voltage, the researchers could also attract the surfactant molecules away from the surface, making the droplet return to its original, flattened shape. The electrodewetting mechanism uses less than 5 volts—as little as 2% of the voltage used in current technologies.
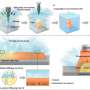
The researchers demonstrated that the process could be used to separate out individual droplets from a larger drop of water, then moved around, split up and merged back together—the four basic operations in digital microfluidics.
They tested the approach with water as well as several solvents and buffer solutions commonly used in chemistry and biology. They also repeated the wetting and dewetting of a water droplet 10,000 times over six hours. In every experiment, the electrodewetting was successful: There were no failures, and the device surface did not show any signs of degradation, even when much higher voltages and currents were used.
More information: Jia Li et al. Ionic-surfactant-mediated electro-dewetting for digital microfluidics, Nature (2019). DOI: 10.1038/s41586-019-1491-x
Journal information: Nature
Provided by University of California, Los Angeles