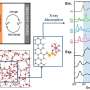
Credit: Saha et al.
Anionic redox chemistry is a fairly new research area that could pave the way for the development of more efficient lithium ion-battery cathode materials such as Li-rich layered oxides. So far, however, anionic redox chemistry approaches have been found to have significant limitations, for instance, leading to voltage fade, large hysteresis and sluggish kinetics.
Researchers at Collège de France and Sorbonne University have carried out a study investigating these limitations, as well as overall challenges that are currently preventing the commercialization of Li-rich layered materials. In their paper, published in Nature Energy, the researchers designed new Li-rich layered sulfides and evaluated their properties.
"This study was aimed at gaining a better understanding of the roadblocks plaguing the commercialization of Li- layered-rich oxides that implicate a new paradigm reactivity involving anionic redox process in addition to cationic ones," Jean-Marie Tarascon , one of the researchers who carried out the study, told TechXplore. "The participation of the anionic redox in sulfides was known for more than 20 years, but was never deeply studied, as its possible advantages were not realized."
Most past studies aimed at achieving a better understanding of Li-rich layered oxides, primarily through cationic substitutions, without changing the material's anion. Tarascon and his colleagues set out to conduct a more thorough investigation in order to better understand the effects of changes in the anionic framework of these materials on their properties and overall performance.
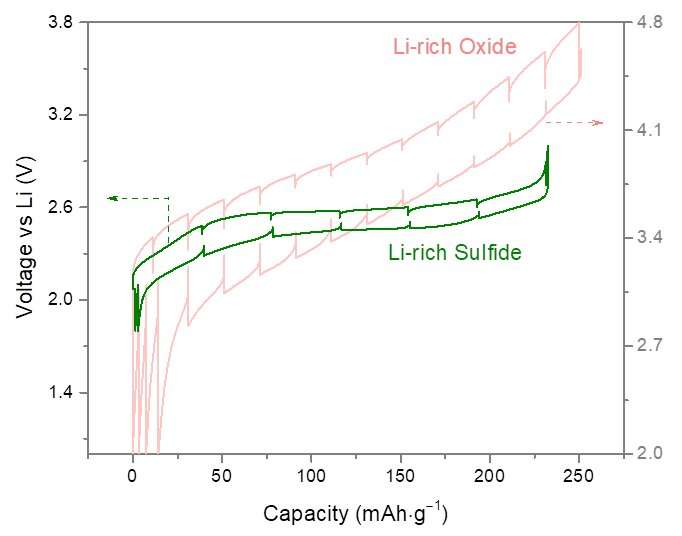
"For having meaningful results and to benchmark Li-rich oxides vs. sulfides we used the same experimental protocol, which entails looking at the voltage hysteresis, voltage decay and energy efficiency," Tarascon explained.
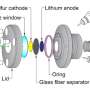
In their study, Tarascon and his colleagues designed new Li-rich layered sulfides Li1.33 – 2y/3Ti0.67 – y/3FeyS2. They found that these sulfides compared favorably to other Li-rich oxide analogues with a negligible initial cycle irreversibility, mitigated voltage fade after long operation cycles, low voltage hysteresis and fast kinetics.
Overall, the researchers observed that shifting from the oxygen ligand to the sulfur ligand alleviates some of the issues associated with anionic redox. However, the substitution also has its disadvantages, as it penalizes the material's redox potential and in turn its energy density.
"We found that voltage hysteresis as well as voltage decay was decreased while energy efficiency was increased, providing evidence that solutions to circumvent the roadblocks associated to Li-rich layered oxides do exist," Tarascon said. "However, there is a price for this, which is a lower energy density due to a lower redox potential."
The study carried out by Tarascon and his colleagues offers valuable insight about the advantages and disadvantages of implementing anionic redox in Li-rich layered sulfides. The observations it collected could ultimately inform the design of new lithium ion-battery cathode materials with higher energy-efficiencies/densities.
"Our future research will be directed towards designing new redox-active Li-rich oxy-sulfide materials for increasing the redox voltage of the material and so that it won't be moisture sensitive," Tarascon said. "This will lead to the development of oxy-sulfides compounds that will have a higher voltage and will be more stable against moisture."
More information: Sujoy Saha et al. Exploring the bottlenecks of anionic redox in Li-rich layered sulfides, Nature Energy (2019). DOI: 10.1038/s41560-019-0493-0
Journal information: Nature Energy
© 2019 Science X Network