Thin radio-frequency detector monitors capacity of commercial lithium-ion battery

A new paper-thin radio-frequency detector designed to work inside a lithium-ion battery provides information about the battery's health while charging and discharging.
"It could enable researchers to check a battery's function and capacity after years of storage without destroying it," said Eric Sorte, a physicist at Sandia National Laboratories.
The work, funded by Sandia's Laboratory Directed Research and Development program will help researchers better understand and characterize batteries to improve them for renewable storage and national security applications. Manufacturers also might use this one day to run diagnostic tests, Sorte said.
Magnetic resonance detector moved inside battery
As a lithium-ion battery powers an electronic device and then recharges, chemical and physical changes happen inside that reduce its function over time. Molecular side products form as lithium ions nestle into and leave each electrode. These molecules can consume the active lithium and reduce a battery's capacity. Electrodes also can undergo unwanted chemical changes, reducing their ability to stay charged. Microscopic spikes of lithium can grow from an electrode surface, consuming key charge-carrying ions and creating potentially flammable conditions.
As researchers work to improve the performance of lithium-ion batteries, they tweak a battery's chemical components and cycle the system through many charges and discharges. Then they open the battery and examine the materials under a microscope to see how their structure and composition have changed.
Researchers could get that information much faster if they could monitor conditions inside a battery as it's charging and discharging. One way they currently do that is with a technique that uses the same principles as magnetic-resonance imaging in hospitals. This method provides clues to a molecule's structure and environment by looking at signals from a specific element in that molecule.
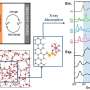
Here's how it works: First, the instrument sends a pulse of radio waves tailored to interact with a specific atomic nucleus in elements such as lithium, sulfur or hydrogen. As a nucleus settles back to its original state, it gives off a signal that changes predictably depending on an atom's surroundings.
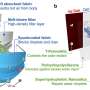
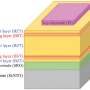
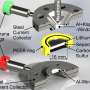
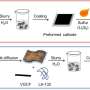
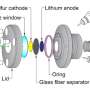
Researchers have used this technique to look at chemical changes in batteries before, but they had to modify battery components in ways that don't exist in working batteries. This new detector, created by Sorte along with Sandia materials scientist Todd Alam and other colleagues, is designed to work in batteries as they are made for everyday use. It is thinner than a sheet of paper and can be made to fit inside a battery of any shape.
The researchers have already slipped it inside a commercially available battery. They imagine one day inserting the detector strip inside a battery during manufacturing, so it already contains the component needed for a quick health check.
Specific signals from molecular components indicate charge, chemical changes
Using this detector, Sorte and his colleagues can see unique signals for lithium ions as they interact with the material in each electrode. This enables them to track how much charge a battery holds throughout repeated charging and discharging cycles; declining capacity is a sign that a battery is dying.
The researchers can also see unique signals from molecules produced during side reactions as a battery operates. They can monitor these molecular side products and then tweak a battery's chemical components to reduce undesirable reactions. These changes can help them improve batteries to have properties needed for applications such as large-scale renewable energy storage. Manufacturers also could use this device one day to run diagnostic tests on batteries, Sorte said.
The same approach and detector strip could be used to look at the inner workings of vanadium flow batteries and other chemistries too, he added. Sorte also is working on monitoring the inner life of a battery using the electrodes already there, so that no additional components are needed.
More information: Eric G. Sorte et al. In Situ Stripline Electrochemical NMR for Batteries, ChemElectroChem (2018). DOI: 10.1002/celc.201800434