March 19, 2020 feature
A cyclic phosphate-based electrolyte for safe and high voltage lithium-ion batteries
In light of the ongoing shift toward renewable energy technologies and the growing number of Internet of Things (IoT) devices, researchers worldwide have been trying to develop batteries that can operate more efficiently and for longer periods of time. Lithium-ion batteries (LIBs) are currently the preferred energy-storage technology for portable electronics, as they contain organic electrolytes, which typically enable high operating voltages and energy densities.
Despite their widespread use, further increasing the performance of existing LIBs could have a significant impact on their safety. In fact, these batteries contain highly volatile and flammable organic carbonates, which, if ignited, can cause considerable damage.
In recent years, researchers have made significant efforts toward overcoming these safety issues, for instance, by using additional substances or by optimizing the materials separating battery components. While some of these strategies successfully reduced the risk of the battery catching fire, as long as LIBs are made with highly flammable electrolytes, accidents may still occur.
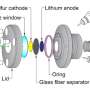
In hope of paving the way for safer and better-performing LIBs, researchers at the University of Tokyo have recently designed and synthesized an alternative cyclic phosphate-based electrolyte that is non-flammable. Their electrolyte, presented in a paper published in Nature Energy, enables safe, highly stable operation and high voltage, outperforming solvents contained in most existing LIBs.
"The electrolyte solvent for lithium-ion batteries (LIBs) has been unchanged for nearly 30 years," Prof. Atsuo Yamada, one of the researchers who developed the new electrolyte, told TechXplore. "We thus think that there should be large room for developing advanced LIBs, if we find an alternative solvent. With this in mind, under the guidance of Prof. Makoto Gonokami, president of the University of Tokyo, we set out a collaboration with Prof. Eiichi Nakamura, who is a highly established researcher in the field of organic synthesis, to design a new electrolyte solvent with the goal of increasing battery performance and safety."
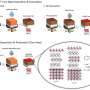
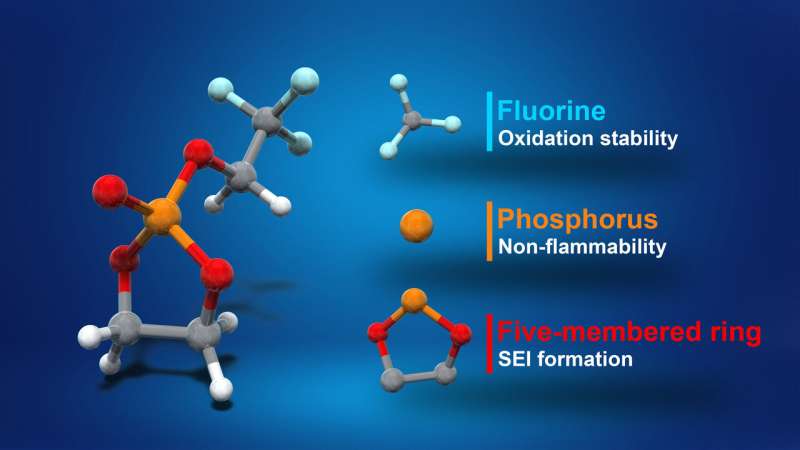
Yamada, Nakamura and their colleagues designed their cyclic phosphate-based electrolyte by fusing the chemical structures of the conventional electrolyte solvent EC and a fire retardant. This gives the phosphate properties of both molecules, including the high-voltage tolerance of the solvent and the non-flammability of the fire retardant, minimizing the risk of LIBs catching fire.
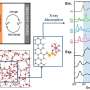
When synthesizing the electrolyte, the researchers found that the most effective formula contained 0.95 M LiN (SO2F)2 in TFEP/2,2,2-trifluoroethyl methyl carbonate. This specific composition enabled the synthesis of an electrolyte with remarkable non-flammability and a self-extinguishing time of zero, as well as stable operation of graphite anodes and high-voltage LiNi0.5Mn1.5O4 cathodes.
"Unexpectedly, the new electrolyte solvent can increase the battery voltage from current 3.8 V to 4.6 V and also improve the battery life," Prof. Yamada said. "We were surprised to see that the designed solvent indeed showed both high-voltage tolerance and fire-retarding ability, as we expected from its chemical structure. Importantly, this is the first case that such a rational design of chemical structures succeeded in battery electrolytes."
Yamada, Nakamura, and their colleagues are among the first to identify an alternative electrolyte solvent that could increase the safety of LIBs while also enhancing their performance. In the future, their cyclic phosphate-based electrolyte could be used to create safe and highly efficient batteries for a wide range of electronic devices.
"We hope that our work will stimulate many researchers to design and develop a variety of new materials for better batteries," Prof. Yamada said. "We now plan to continue working on this new electrolyte solvent for commercial battery applications and develop new multifunctional battery materials based on our design strategy."
More information: Qifeng Zheng et al. A cyclic phosphate-based battery electrolyte for high voltage and safe operation, Nature Energy (2020). DOI: 10.1038/s41560-020-0567-z
yamada-lab.t.u-tokyo.ac.jp/en/
© 2020 Science X Network