A breakthrough in solar cell efficiency

A research group led by Prof. Chen Tao and Prof. Zhu Changfei, and their collaborator Prof. Hao Xiaojing at UNSW, developed a hydrothermal deposition method for the synthesis of antimony selenosulfide for solar cell applications. With this absorber material, the solar cell breaks the 10% benchmark efficiency barrier. This result has been published in Nature Energy entitled "Hydrothermal deposition of antimony selenosulfide thin films enables solar cells with 10% efficiency."
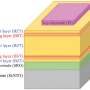
Antimony selenosulfide, Sb2(S,Se)3, as a ROHS-compliant and earth-abundant light harvesting material, has received increasing interest during the past few years. The band gap of Sb2(S,Se)3 is tunable in the range of 1.1-1.7 eV, satisfying the requirement for optimal sunlight harvesting. In addition, Sb2(S,Se)3 possesses a high extinction coefficient, and film thickness of about 500 nanometers can absorb sufficient light irradiation. With these advantages, Sb2(S,Se)3 is a promising energy material for the applications of lightweight and portable electricity generation devices.
Considering that Sb2(S,Se)3 consists of earth-abundant elements and has excellent stability, the improvement in breaking the 10% efficiency benchmark will break ground for a commercialization path. In this study, the authors found that the hydrothermal deposition at supercritical conditions enables the generation of compact and flat film with homogeneous element distribution in the lateral direction. These superior characteristics allow the efficient carrier transport and suppression of detrimental recombination. With further optimizations of the band gap, cation/anion ratio, crystal orientation and defect properties, the device successfully achieves a record power conversion efficiency.
A reviewer of this paper praised the work, commenting that "This paper presents a landmark efficiency value for Sb2(S,Se)3 solar cells breaking the 10% barrier. This achievement sheds new light on the investigation and application of Sb2(S,Se)3 ...".
More information: Rongfeng Tang et al. Hydrothermal deposition of antimony selenosulfide thin films enables solar cells with 10% efficiency, Nature Energy (2020). DOI: 10.1038/s41560-020-0652-3