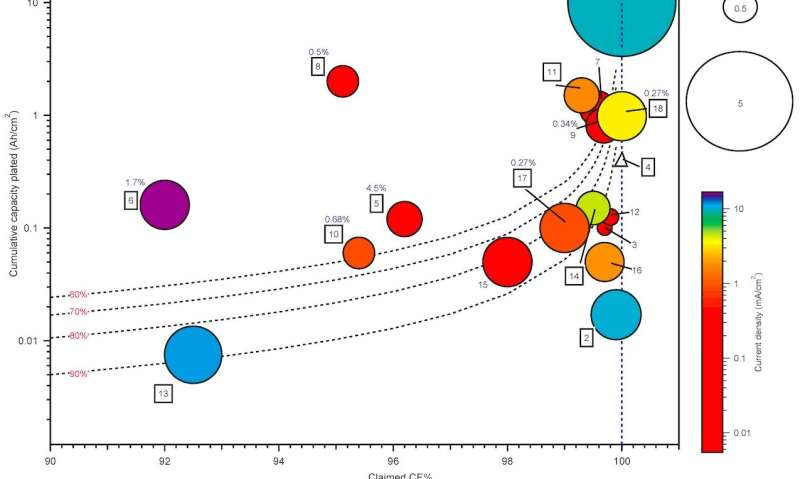
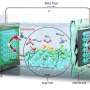
Summary of published Zn plating/stripping CE measurements. Credit: Ma et al. (Nature Energy, 2020).
Rechargeable zinc metal batteries (RZMBs) could be a viable alternative to lithium ion batteries, which are currently used to power many types of electronic devices, as well as electric vehicles. Recent studies suggest that using optimized electrolytes could lead to RZMBs with highly reversible zinc plating/stripping, achieving Coulombic efficiencies (CEs) close to 100%.
Nonetheless, as teams who set out to design optimized electrolytes for RZMBs used different experimental methods, comparing existing electrolytes and identifying the most efficient ones has so far proved challenging. In a recent paper published in Nature Energy, researchers at the U.S. Army Research Laboratory (ARL) review recent progress in the development of these electrolytes, outlining a set of protocols that could be used to evaluate them and compare their performance. The lead author of this work is Lin Ma, a distinguished postdoctoral fellow at ARL.
"Our research focuses on developing transformative materials that will enable next-generation batteries for both consumer and army energy storage applications," Mashall A. Schroeder and Kang Xu, two of the researchers who carried out the study, told TechXplore via email. "After joining the Department of Energy's Joint Center for Energy Storage Research (JCESR 2.0) in the fall of 2018, we took a closer look at multivalent-ion (Mg2+, Ca2+, Zn2+, Al3+, etc.) battery systems, which promise to offer unique benefits compared to Li but at the cost of addressing major fundamental challenges."
Ma, Schroeder, Xu and their colleagues have so far decided to focus their research efforts on rechargeable batteries with zinc metal anodes. These batteries compare favorably to lithium-based batteries, as they present a significantly higher energy per volume (5854 AhL-1) and a high energy per weight (820 mAhg-1). In addition, zinc metal is far more abundant than lithium, as there are larger amounts of it in the Earth's crust.
Despite their advantageous characteristics, so far, very few companies have considered producing RZMBs on a large scale due to the poor cycling reversibility of the zinc metal anode when the batteries are used in real-world settings. Overall, past studies evaluating RZMBs produced mixed and sometimes conflicting results and thus do not offer enough evidence to back their commercialization.
The main objective of the study by Ma, Schroeder, Xu and their colleagues was to paint a clearer picture of recent advancements in the development of RZMBs, while also proposing the use of standardized protocols that could help to ascertain the value of this new battery technology in the future. In their paper, the researchers introduce commercial targets and outline a set of rigorous methodologies that could be used to evaluate new RZMBs.
"The primary figure of merit for a rechargeable battery system is the Coulombic efficiency (CE, %), which is a relation between the total charge that can be extracted from a battery compared to the total charge put into a battery over a full cycle," Schroeder and Xu explained. "It is a critical measure of reversibility that suggests whether a battery can be rechargeable and for how many cycles. In RZMBs, this is primarily determined by how reversibly zinc can be electrochemically plated and stripped, but this behavior often critically depends on the experimental conditions adopted by researchers."
In their paper, Ma, Schroeder and Xu highlight a number of experimental conditions that can affect the reversibility of zinc, including how quickly/slowly it is cycled (i.e., current density—mA cm-2) and the amount of zinc plated/stripped per cycle (areal capacity—mAh cm-2). In addition, when reviewing past works evaluating the effectiveness of electrolyte systems or approaches for improving the performance of RZMBs, they found that many teams had performed tests using inconsistent or unrealistic values for current densities and areal capacities.
"After assembling the first comprehensive comparison of the ~20 published studies on this topic, we discovered significant variability in published test methods and parameters with poor alignment toward commercial targets for zinc metal batteries," Schroeder and Xu said. "This leads to confusion in assessing the relative performance and commercial viability of these systems."
The recent paper introduces a valuable set of standard protocols that could be used to evaluate RZMBs more reliably, determining their true Coulombic efficiencies. In the future, these protocols could serve as a guide for teams trying to develop RZMBs and test their zinc reversibility, potentially enhancing coherence and alignment between different research efforts.
"Batteries are complicated electrochemical systems, and there are multiple approaches to improving performance including the use of advanced electrolytes, surface treatments, and novel electrode architectures," Schroeder and Xu said. "Pending adoption by the field, the conclusions and recommended methods from this Perspective have the potential to unify development efforts for zinc metal batteries, streamlining commercialization of this promising system and eventually enabling access to a potentially safer, more sustainable battery technology than Li-based systems with improved energy per volume."
Ma, Schroeder, Xu and their colleagues now plan to continue developing advanced aqueous (i.e., water-based) and non-aqueous electrolytes for next generation battery technologies. As part of this process, they are also trying to attain a detailed and fundamental understanding of the factors impacting the reversibility of RZMBs, in order to identify approaches that can improve their performance.
More information: Lin Ma et al. Realizing high zinc reversibility in rechargeable batteries, Nature Energy (2020). DOI: 10.1038/s41560-020-0674-x
Journal information: Nature Energy
© 2020 Science X Network