The continuing quest to find a better battery
Ten years ago, if you needed a battery for something more than a flashlight or an alarm clock, your options were pretty limited. Lithium-ion batteries had found their way into consumer electronics in the 1990s, and researchers were just beginning to explore their potential for certain automotive applications.
Even in 2010, however, scientists and policymakers knew that lithium-ion batteries would not be the only solution to the energy storage challenge. Different applications, from heavy-duty trucking to electric airplanes to storing intermittently produced renewable energy in large-scale installations on the electric grid, required scientists to think more broadly in terms of chemistry and technology.
To discover pivotal battery technologies beyond lithium-ion, the U.S. Department of Energy (DOE) started the Joint Center for Energy Storage Research (JCESR), an Energy Innovation Hub led by DOE's Argonne National Laboratory designed to bring laboratories, academia, and industry together to solve key challenges in energy storage. Initially designed as a five-year effort, JCESR was tasked with developing batteries that would store dramatically more energy in the same amount of weight while costing far less.
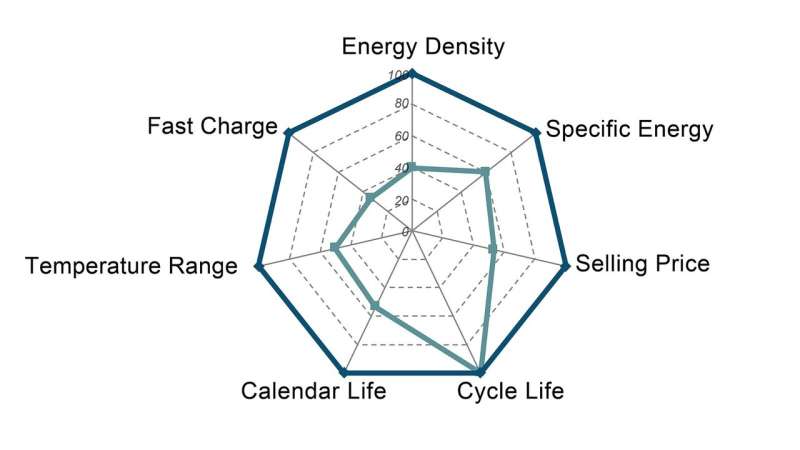
In JCESR's early days, this challenge motivated a search for a "magic bullet"—a battery that, because it met certain energy density and cost requirements, would satisfy the needs of a wide variety of applications. "Initially we thought that we could find a single chemistry, or maybe a family of chemistries, that could work across the board," said JCESR director and Argonne Distinguished Fellow George Crabtree. "We came to see that no matter what battery we ended up designing, a one-size-fits-all approach was never going to truly work."
One of the reasons for this, Crabtree explained, is that different kinds of battery applications require different sorts of tradeoffs. A battery that would be good for an airplane would need to be able to store and produce an exceptionally large amount of energy—several times that of lithium-ion—while being extremely safe. On the other hand, cost would not be a significant factor, he said, because airline operating costs dwarf their capital costs.
Conversely, batteries for cars and light-duty trucks only need to store enough energy to drive several hundred miles before recharging, but they ideally need to be able to be charged quickly, as well as being safe and low-cost.
"It quickly became apparent that you're never going to have one battery that gives you literally everything," said Argonne materials scientist and JCESR research integration lead Lynn Trahey. "That vision worked well for personal electronics, the primary use of lithium-ion batteries until about 2010. Today's world is diverging into many very different applications, and we need a diversity of batteries to serve them."
In JCESR's initial five-year scope, researchers used economic models of different batteries and their applications to inform their experiments and analyses. Aside from consumer electronics—a market whose needs are fairly well-satisfied by lithium-ion technology—JCESR's scientists took the broadest view possible of how groundbreaking energy storage technology could transform the American economy and our way of life.
Batteries for transportation and batteries for the electric grid diverge considerably in their needs. In applications that seek to store large quantities of clean energy on the electric grid for times when the sun isn't shining or the wind isn't blowing, scientists can use large and inexpensive batteries unlike those that would be suitable for an electric car.
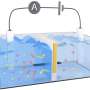
For this grid storage, JCESR scientists investigated the use of redox flow batteries, which unlike lithium batteries use liquid instead of solid electrodes. Redox flow batteries get their name from the fact that a flowing liquid causes one side of the battery to electrochemically reduce, or become more negative, while the other side electrochemically oxidizes, or becomes more positive, as the battery is charged or discharged.
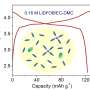
The basic concept of a redox flow battery is fairly simple—each of two differently charged fluids circulate between two tanks, while sharing ions across a transport membrane in the electrochemical cell located between the two sets of tanks. Because the fluids are typically inorganic salts in water-based solutions, however, their energy density and cost saving potential are limited, Crabtree said.
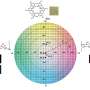
To overcome these limitations with redox flow batteries, JCESR scientists looked to see if they could use organic molecules as the foundation. In doing so, they developed a set of molecules called "redoxmers," which are organic polymers that offer the availability of increased cell voltages when used in organic solvents.
"The wonderful thing about redoxmers is that they are so numerous and so versatile," Trahey said. "There are thousands of different organic molecules that can be configured in thousands of different ways. As we explore this vast space, we learn which configurations enable the targeted performance that we want, such as high voltage, fast charging, high safety or low cost." Machine learning, where computers learn the atomic and molecular features that produce high battery performance, dramatically speeds up the discovery of new and better materials.
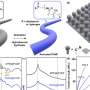
Just as for the grid, batteries for transportation must serve a variety of applications. JCESR scientists have worked on lithium-oxygen, lithium-sulfur and "multivalent ion" batteries. Unlike lithium ions, which have a charge of +1, multivalent ions have a charge of +2 or +3, enabling them to do more electrochemical work each trip through the battery. These batteries all promise much higher energy density at lower cost than lithium-ion, provided the right combination of materials can be found.
A key lesson of JCESR's early years is the challenge of finding the right materials. Although there are many candidates, there are also many performance requirements and many unwanted side reactions that often prevent a promising candidate from making the grade.
In recent years, JCESR's operating philosophy has changed. Instead of trying to find battery chemistries specifically tailored to particular technological needs, JCESR researchers have traded a "top-down" process for a "bottom-up" approach in which they focus on fundamental investigations of different chemical processes on the atomic and molecular levels.
"The idea is to understand the materials and phenomena of energy storage so well that we can design a variety of batteries for a variety of applications," Crabtree said. "An extensive knowledge of energy storage at the atomic and molecular level is the new feature that allows us to custom design a battery atom-by-atom and molecule-by-molecule, and hit the overall battery performance requirements for any application."
"With state-of-the-art techniques such as advanced light sources and machine learning, we can explore areas of battery chemistry that we've never anticipated. That is exciting," Trahey added.