Photo of the operando x-ray studies being performed at a Synchrotron facility. Credit: WMG, University of Warwick
High energy storage batteries for EVs need high capacity battery cathodes. New lithium-excess manganese-rich cathodes are expected to replace existing nickel-rich cathodes but understanding how the manganese and oxygen accommodate charge storage at high voltages is critical for their successful adaption. Research led by WMG, University of Warwick in collaboration with U.S. researchers employed a range of X-ray studies to determine that the oxygen ions are facilitating the charge storage rather than the manganese ions.
Electric vehicles will one day dominate UK roads and are critical for eliminating CO2 emissions, but a major issue car manufacturers face is how to make an affordable long-lasting energy-dense battery that can be charged quickly and efficiently. There is therefore a race to make EV batteries with an energy storage target of 500 Wh/Kg, but these targets are not possible without changing to new cathode materials.
Although progress has continued over the last 10 years to push the performance of state-of-the-art nickel-rich cathodes for EV, the material is unable to provide the energy density needed. To increase the capacity more lithium needs to be used, which means going beyond the ability of nickel to store electron charge.
Lithium-excess manganese-rich cathodes offer sufficient energy density but to reach ultimately reach energy storage targets of 500Wh/Kg we need to understand how the electron charge is stored in the material. Simply put, is the electron charge stored on the manganese or oxygen sites.
In the paper, "Whither Mn Oxidation in Mn-Rich Alkali-Excess Cathodes?", published in the Journal ACS Energy Letters today the 17th of February, researchers from WMG, University of Warwick have overcome a significant milestone in understanding of charge storage in lithium-excess manganese-rich cathodes.
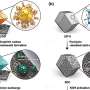
Li-excess compounds that involve conventional and non-conventional redox, conventional refers to metal ions changing their electron density. Reversibly changing the electron density on the oxygen (or oxygen redox) without it forming O2 gas is unconventional redox. Various computational models exist in the literature describing different mechanisms involving both, but careful X-ray studies performed while the battery is cycling (operando) are ultimately required to validate these models.
Researchers between the UK and US, led by WMG at the University of Warwick, performed operando X-ray studies to precisely quantify manganese and oxygen species at high voltages. They demonstrated how X-ray beams could irreversibly drive highly oxidized manganese (Mn7+) to trapped O2 gas irreversibly in other materials.
However, by performing careful operando X-ray studies that circumvented beam damage and observe only trace amounts of Mn7+ forming upon charging in Li-excess cathodes during battery cycling.
Professor Louis Piper, from WMG, University of Warwick explains, "We have ultimately resolved that oxygen rather than metal redox is driving the higher capacity, which means we can now design better strategies to improve cycling and performance for this class of materials."
More information: Mateusz Jan Zuba et al. Whither Mn Oxidation in Mn-Rich Alkali-Excess Cathodes? ACS Energy Lett. 2021, 6, XXX, 1055–1064 February 17, 2021 doi.org/10.1021/acsenergylett.0c02418
Provided by University of Warwick