April 30, 2021 report
A zero-gap gas diffusion electrode-based electrolyzer with improved stability
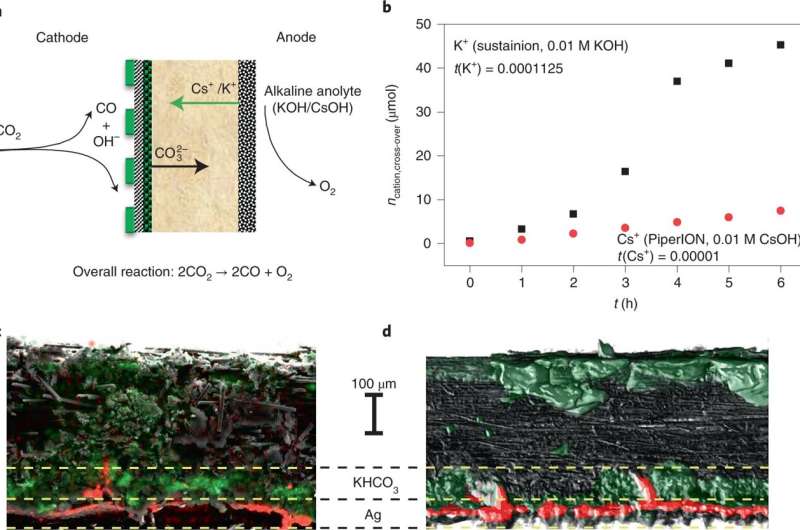
A team of researchers from the University of Szeged has developed a zero-gap GDE-based electrolyser that has better stability than others in its category. In their paper published in the journal Nature Energy, the group describes their work, which involved attempting to improve the prospects of using electrolysers as a means for creating fuel sources. Klaas Jan P. Schouten with the University of Amsterdam has published a News & Views piece describing the work in the same journal issue.
As Schouten notes, most of the materials that are currently used to make fuels are carbon-based, which, of course, has led to problems with pollution and global warming. So scientists have begun to look for other ways to make them. One approach is the use of electrolysers to convert carbon dioxide to a desired fuel. He notes that water-fed electrolysers allow for producing both chemicals and fuels using only carbon dioxide, water and a source of renewable energy. He also notes that there are four main ways of using electrolysers for such applications, but only one of them has both high selectivity and high efficiency—one based on gas diffusion electrodes (GDEs). Unfortunately, prior attempts to produce fuel using GDEs have been hampered by cathode precipitate formation, which reduces stability.
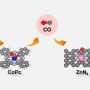
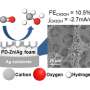
In this new effort, the team in Hungary has found a way to sidestep that problem, and in so doing, has created an electrolyzer that is more stable than prior efforts. They used it to create carbon monoxide as a fuel. The technique involves using a silver carbon dioxide reduction catalyst dispersed on the carbon dioxide—it was pushed against an anion exchange membrane. Also, the anode was made by coating titanium with IrOx placed against the other end of the membrane. The researchers call the result an operando activation and regeneration process. The carbon dioxide reduction to carbon monoxide was conducted by applying electric current.
Over a 200-hour run, the approach proved capable of producing carbon monoxide matching other techniques based on alkaline electrolytes. The researchers also tried their approach with five kinds of electrolytes and three kinds of membranes, and found it worked just as well. They suggest their approach could be easily scaled up to meet manufacturing needs.
More information: B. Endrődi et al. Operando cathode activation with alkali metal cations for high current density operation of water-fed zero-gap carbon dioxide electrolysers, Nature Energy (2021). DOI: 10.1038/s41560-021-00813-w
© 2021 Science X Network