We've smelted a billion tons of recyclable aluminum. Do we need to make more?

Aluminum is light and versatile, but massively energy-intensive to produce, requiring 10% of Australia's entire electricity output . Recycling it uses just a fraction of the energy. Why aren't we closing the loop?
This metal—the most abundant in the Earth's crust—is used in everything from kitchen utensils to soft drink cans, buildings and plane parts.
Since we discovered how to extract it in the 19th century, around 1 billion tons of aluminum has been smelted. Of that, three quarters is accessible for recycling.
Unfortunately, aluminum's energy-intensive production has major consequences for climate change. We must power aluminum production with renewables, and find better ways to recycle this most useful metal.
To provoke thought about aluminum and its energy needs, I collaborated with designer Kyoko Hashimoto to produce new works of design using aluminum. These mirrors and vases are currently on display as part of the National Gallery of Victoria's Sampling the Future exhibition.
As critical designers, we hope to communicate the waste problem created by mixing aluminum into unrecoverable composites, and reframe the perception of the metal's value, which has diminished since its discovery.
From precious metal to common disposable
When aluminum was first extracted and purified, it was more expensive than gold. Napoleon III famously had his son's baby rattle made from aluminum. In 1884, as the most exotic metal of its day, it was used for the pyramid cap on the Washington monument.
Now, aluminum is plentiful and cheap. Australia is the world's leading producer of the main ore, bauxite, and we export most of it for processing overseas.
Impressively large amounts of energy are needed to break the tight bonds of the metal from its oxides. In Australia, making new aluminum represents 6.5% of our greenhouse gas emissions. The intense chemical process also creates toxic byproducts and pollution.
Over the past few years, aluminum production has shifted to countries such as Iceland, with cheap and sustainable energy from geothermal sources.
Unfortunately, the lion's share of production takes place in countries such as China, and often relies on Australian coal. Australia also ranks high in CO₂ emissions from alumina refining, an intermediate stage of processing.
Recycling aluminum requires only around 5%of the energy of smelting, the highest recycling energy saving for any major material.
Global aluminum recycling rates range from 34% to 70%. In Australia, recycling rates for aluminum packaging are between 44% to 66%, but likely lower across industrial and consumer products.
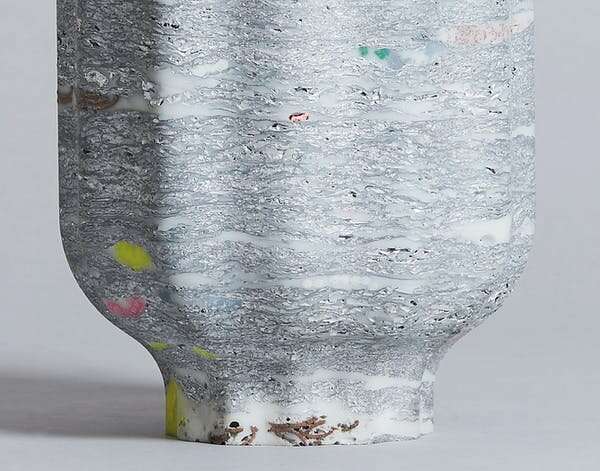
Why don't we recycle all our aluminum?
There is scope to boost recycling, but product design and waste streams pose challenges.
For example, the aluminum we used in our designs is newly milled "5083," a high grade, corrosion-resistant magnesium alloy with traces of manganese and chromium. Such trace metals are used to improve rigidity, corrosion resistance or welding capacity.
While our supplier sends offcuts and scrap for recycling, the mix of different alloys means these are 'downcycled' into lower-grade products. Most of Australia's aluminum scrap is exported, so increasing our local recycling would decrease the emissions from shipping this scrap offshore.
There are losses across industrial and consumer waste streams alike, despite new sorting technologies. Magnetic eddy current technologies can sort metal objects from non-metal objects and even non-ferrous metal objects from each other.
The job gets harder when you encounter multi-material objects. Metal fasteners like screws, rivets and pins, as well as bonded adhesives, are leading causes of impurities in aluminum recycling.
Many aluminum products are designed also as "monstrous hybrid" composites using materials unable to be easily separated. Coffee pods are the most famous example.
These problems have to be fixed at the design stage. Such issues mean aluminum is steadily lost to human use, ending up in landfill and back into the environment.
While aluminum ores are readily found across the world, the metal is curiously absent from biological systems. It has had little role in plant or animal evolution and biologically available aluminum can be toxic. We do not know if this will have long-term consequences in nature.
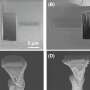
We drew attention to these hidden issues in the design of our "metalloplastiglomerate" vases. They were made by crumpling and hammering aluminum sheet around organic fiber, plastic and soft metal waste.
In these works, we speculate about what will happen to aluminum as it is ejected from collapsing cities and transforms back into geological rock in the far future.
Could we pioneer a circular economy with aluminum?
Even as the world fights to stave off dangerous climate change, demand for new aluminum is estimated to double or triple by 2050. If Australia's aluminum recycling improves, we're likely to keep making new aluminum to supply increasing international demand.

Australia exports most of its new aluminum, despite our smelters relying on heavy government subsidies. These smelters have been used by politicians to justify power from fossil fuels for their baseload output.
This is a furphy. Hydroelectric power works well with smelters too. Aluminum production using renewable energy may be justified in Australia, if we can manage its other environmental impacts.
Australia should also stop exporting bauxite or alumina to countries with fossil fuel powered smelters.
It's entirely possible to end the need for new aluminum. Since we discovered the metal, we have produced around 1 billion tons of it. Around 75% is in current use, and available for recycling as it becomes necessary. Planning to stop producing new aluminum would create an incentive to better care for the metal we have and reduce waste.
And while aluminum is prized as a light and strong material, there are other materials with potential to replace it, including those that capture carbon instead of release it.
Slowing and eventually stopping new aluminum production would demonstrate how the world's economy can thrive under degrowth—a controlled contraction of production to stem climate change and function within the planet's ecological limits.
We considered this idea in the design of our aluminum and bauxite mirrors. They contain roughly the amount of aluminum able to be produced from the bauxite rocks that hold them. To communicate a sense of conservation, we modified the rock as little as possible. We made one cut to expose its beautiful pebble-like internal structure, and a second to hold the mirror.
In our designs, we hope to show the technological beauty of aluminum production, as well as the care with which we should approach it.
Aluminum's unique properties drive ever greater production. But a growth at all costs mentality for resource extraction is perilous—especially when we can use what we already have.
This article is republished from The Conversation under a Creative Commons license. Read the original article.![]()