Neutron scattering study points the way to more powerful lithium batteries
An international team of scientists has found a way to improve battery design that could produce safer, more powerful lithium batteries.
Apr 16, 2024
0
25
Energy & Green Tech

An international team of scientists has found a way to improve battery design that could produce safer, more powerful lithium batteries.
Apr 16, 2024
0
25
Energy & Green Tech
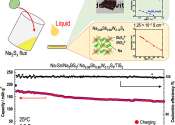
The pursuit of greener energy also requires efficient rechargeable batteries to store that energy. While lithium-ion batteries are currently the most widely used, all-solid-state sodium batteries are attracting attention ...
Apr 11, 2024
0
126
Energy & Green Tech

A study led by Professor Sung-Kyun Jung and his research team in the School of Energy and Chemical Engineering at UNIST has unveiled a more stable approach to utilizing all-solid-state batteries (ASSBs), setting a new standard ...
Apr 9, 2024
0
27
Energy & Green Tech

Scientists have discovered a stable and highly conductive lithium-ion conductor for use as solid electrolytes for solid-state lithium-ion batteries.
Apr 2, 2024
0
71
Electronics & Semiconductors

Lithium-ion batteries (LIBs), mainly used as the power of computer, communication and consumer electronic products, require higher energy density, longer cycling life, faster-charging capability, and a broader operating temperature ...
7 hours ago
0
13
Energy & Green Tech

Oxide ion conductors used in solid-state fuel cells often fail to reach full potential when operating at temperatures below 500 oC, but researchers from Tokyo Tech have recently found a solution to this problem. They demonstrated ...
Apr 10, 2024
0
10
Energy & Green Tech

Korea Electrotechnology Research Institute (KERI) has reached a significant milestone with a study published in Energy Storage Materials, marking a crucial stride toward the commercialization of all-solid-state batteries, ...
Mar 19, 2024
0
1
In chemistry, an electrolyte is any substance containing free ions that make the substance electrically conductive. The most typical electrolyte is an ionic solution, but molten electrolytes and solid electrolytes are also possible.
Commonly, electrolytes are solutions of acids, bases or salts. Furthermore, some gases may act as electrolytes under conditions of high temperature or low pressure. Electrolyte solutions can also result from the dissolution of some biological (e.g., DNA, polypeptides) and synthetic polymers (e.g., polystyrene sulfonate), termed polyelectrolytes, which contain charged functional groups.
Electrolyte solutions are normally formed when a salt is placed into a solvent such as water and the individual components dissociate due to the thermodynamic interactions between solvent and solute molecules, in a process called solvation. For example, when table salt, NaCl, is placed in water, the salt (a solid) dissolves into its component ions, according to the dissociation reaction
It is also possible for substances to react with water producing ions, e.g., carbon dioxide gas dissolves in water to produce a solution which contains hydronium, carbonate, and hydrogen carbonate ions.
Note that molten salts can be electrolytes as well. For instance, when sodium chloride is molten, the liquid conducts electricity.
An electrolyte in a solution may be described as concentrated if it has a high concentration of ions, or dilute if it has a low concentration. If a high proportion of the solute dissociates to form free ions, the electrolyte is strong; if most of the solute does not dissociate, the electrolyte is weak. The properties of electrolytes may be exploited using electrolysis to extract constituent elements and compounds contained within the solution.
This text uses material from Wikipedia, licensed under CC BY-SA