EU lawmakers impose single charger for all smartphones
The EU parliament on Tuesday passed a new law requiring USB-C to be the single charger standard for all new smartphones, tablets and cameras from late 2024.
Oct 4, 2022
3
220
Consumer & Gadgets

The EU parliament on Tuesday passed a new law requiring USB-C to be the single charger standard for all new smartphones, tablets and cameras from late 2024.
Oct 4, 2022
3
220
Engineering

Engineers at Tufts University have created a first-of-its-kind flexible electronic sensing patch that can be sewn into clothing to analyze your sweat for multiple markers. The patch could be used to to diagnose and monitor ...
Jul 28, 2020
0
125
Internet

Tim Hart was sitting on his couch one evening in November 2011 when he got an email with the subject line: "I'm watching." The message that followed was short and to the point …
Feb 26, 2021
0
10
Electronics & Semiconductors
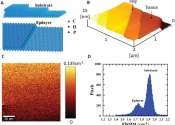
A National Institute for Materials Science (NIMS) research team has developed the world's first n-channel diamond MOSFET (metal-oxide-semiconductor field-effect transistor). The developed n-channel diamond MOSFET provides ...
Mar 15, 2024
0
103
Electronics & Semiconductors

Researchers at the Department of Instrumentation and Applied Physics (IAP), Indian Institute of Science (IISc), have designed a novel ultramicro supercapacitor, a tiny device capable of storing an enormous amount of electric ...
Mar 31, 2023
0
3170
Electronics & Semiconductors

For decades, electronics engineers have been trying to develop increasingly advanced devices that can perform complex computations faster and consuming less energy. This has become even more salient after the advent of artificial ...
Engineering

Researchers at the University of Colorado Boulder have developed a new, low-cost wearable device that transforms the human body into a biological battery.
Feb 10, 2021
3
1499
Engineering

A new kind of solar panel, developed at the University of Michigan, has achieved 9% efficiency in converting water into hydrogen and oxygen—mimicking a crucial step in natural photosynthesis. Outdoors, it represents a major ...
Jan 4, 2023
2
326
Engineering

The 3D printing company Desktop Metal has just announced the release of Forust, a new tool using wood to 3D print objects. The company, founded in 2019, focuses on 3D printing for interior design. With printing methods deemed ...
Electronics & Semiconductors

In recent decades, machine learning and deep learning algorithms have become increasingly advanced, so much so that they are now being introduced in a variety of real-world settings. In recent years, some computer scientists ...
The electron is a subatomic particle that carries a negative electric charge. It has no known substructure and is believed to be a point particle. An electron has a mass that is approximately 1836 times less than that of the proton. The intrinsic angular momentum (spin) of the electron is a half integer value of 1/2, which means that it is a fermion. The anti-particle of the electron is called the positron, which is identical to electron except that it carries electrical and other charges of the opposite sign. In collisions electrons and positrons annihilate, producing a pair (or more) of gamma ray photons. Electrons participate in gravitational, electromagnetic and weak interactions.
The concept of an indivisible amount of electric charge was theorized to explain the chemical properties of atoms, beginning in 1838 by British natural philosopher Richard Laming; the name electron was introduced for this charge in 1894 by Irish physicist George Johnstone Stoney. The electron was identified as a particle in 1897 by J. J. Thomson and his team of British physicists. Electrons are identical particles that belong to the first generation of the lepton particle family. Electrons have quantum mechanical properties of both a particle and a wave, so they can collide with other particles and be diffracted like light. Each electron occupies a quantum state that describes its random behavior upon measuring a physical parameter, such as its energy or spin orientation. Because an electron is a type of fermion, no two electrons can occupy the same quantum state; this property is known as the Pauli exclusion principle.
In many physical phenomena, such as electricity, magnetism, and thermal conductivity, electrons play an essential role. An electron generates a magnetic field while moving, and it is deflected by external magnetic fields. When an electron is accelerated, it can absorb or radiate energy in the form of photons. Electrons, together with atomic nuclei made of protons and neutrons, make up atoms. However, electrons contribute less than 0.06% to an atom's total mass. The attractive Coulomb force between an electron and a proton causes electrons to be bound into atoms. The exchange or sharing of the electrons between two or more atoms is the main cause of chemical bonding.
Electrons were created by the Big Bang, and they are lost in stellar nucleosynthesis processes. Electrons are produced by cosmic rays entering the atmosphere and are predicted to be created by Hawking radiation at the event horizon of a black hole. Radioactive isotopes can release an electron from an atomic nucleus as a result of negative beta decay. Laboratory instruments are capable of containing and observing individual electrons, while telescopes can detect electron plasma by its energy emission. Electrons have multiple applications, including welding, cathode ray tubes, electron microscopes, radiation therapy, lasers and particle accelerators.
This text uses material from Wikipedia, licensed under CC BY-SA