Credit: AFMD Group
Organic solar cells have the potential to become a source of renewable energy which is inexpensive and fast to roll out and scale up. Physicists at the University of Oxford have explored some of the scientific fundamentals of how these cells form and perform.
Solar energy currently accounts for less than 2 % of electricity generated worldwide but could make a big contribution to sustainability. Achieving scale involves deploying it over a large surface area. "We need several thousand square kilometres to make a dent in the world's energy supply so the ability to scale up fast and at low cost is vital," says Professor Moritz Riede, the main researcher for OSC Go and associate professor of soft functional nanomaterials at Oxford. "You would like to be able to coat square kilometres with solar cells in an inexpensive and fast way."
Most commercially-available solar systems are based on inorganic silicon semiconductors. Organic, carbon-based photovoltaics could offer plenty of advantages – they are lightweight and flexible, can come in different colours, and are made cheaply, using low temperature processes. Unfortunately, currently they are also a lot less efficient at converting sunlight into electricity than conventional silicon-based systems.
The OSC Go team has spent the past four years exploring some of the fundamental questions about how organic solar cells (OSCs) are made with a view to improving their performance.
Observing deposition
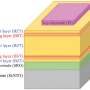
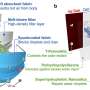
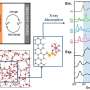
Structure-property relationships have been a major focus of their research as the arrangement of the molecules inside an organic solar cell can have a big impact on its performance. The team devised ways of using light of various wavelengths – from X-rays to near infrared – to probe how the molecules arrange themselves in thin films. "This is usually investigated when it is fully formed, after the process is over, but we can watch the molecules during the deposition process," says Prof. Riede, "so we can see how the molecules pack and what we can do to manipulate their arrangement."
Using Fullerene C60, a material often used to make OSCs, the team managed to observe how defects can form in these thin films and even influence the outcome. "We have observed C-stacking faults in a particular molecular direction," says Prof. Riede, "that gave us an important data point on the structural level for interpreting how such devices perform."
Great role models
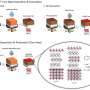
In an organic solar cell, sunlight is absorbed in the photoactive layers usually consisting of a mix of two materials – electron donors and acceptor molecules – where it is converted it into electricity. The OSC Go researchers spent time evaluating the performance of dilute heterojunction solar cells – ones where the donor content is 5 % or less.
"These devices were found to work surprisingly well," says Prof. Riede, "so we have been looking in pure C60 cells to see how the molecules pack and how they pack and perform in the presence of other molecules. These devices are excellent model systems and we tried to join the microstructural results with photo-physical ones."
The effect of changes in the microstructure on the performance of devices was a third area of study. In collaboration with chemical company Merck, the team investigated what happens when the OSC film is subjected to high temperatures or exposed to sunlight for long periods of time, which they will during operation. "We measured the changes in microstructure with X-rays and other methods and were able to relate these changes to changes in the performance of OSCs," says Prof. Riede, "so this allows us to look for ways to inhibit that."
A better understanding of what is going on at the nanoscale will be very useful when it comes to choosing which materials to use for making efficient OSCs, Prof. Riede believes.
"There is a plethora of materials you can use and one can tune the materials and improve their performance by clever chemical design and good manufacturing conditions," he says, "but to be able to do so, you also have to be able to understand the fundamentals."
Provided by CORDIS