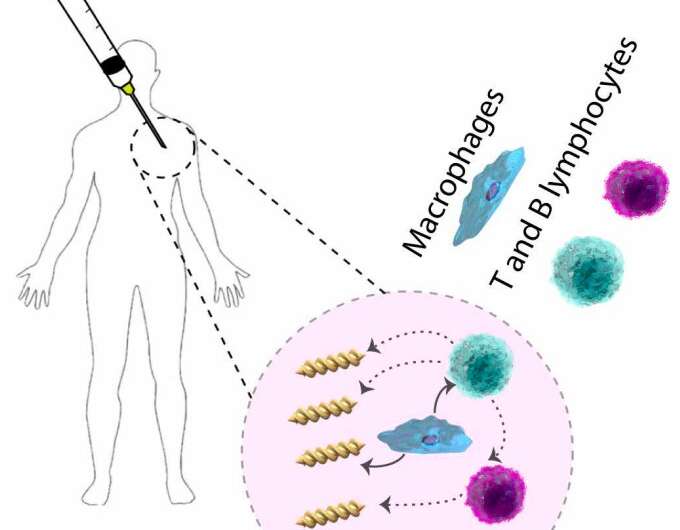
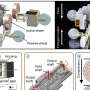
Figure summarizing the interactions between microswimmers and the human immune system. Credit: Yasa et al.
Over the past few years, robotics researchers have designed tiny and untethered swimming robots, also known as microswimmers, with increasingly advanced sensing and locomotion capabilities. These microrobots could prove very useful in medical settings, particularly for the implementation of minimally invasive targeted therapies in parts of the body that are difficult to reach, such as the central nervous system or vascular system.
While microswimmers could help to intervene in sensitive sites inside the body, their use also comes with some risks. For instance, they could elicit undesirable responses from the human immune system, which may perceive them as intruders.
The immune system is designed to recognize and protect the body from external or foreign objects and organism. When cells of the immune system, such as macrophages, detect a foreign object or intruder, they act by trying to eliminate or neutralize it via a process known as phagocytosis.
In order to circumvent this issue and ensure that microrobots do not incite immune responses, the robots should be designed carefully, in ways that minimize their physical interaction with cells of the immune system. Those designing them should pay particular attention to their shape, structure and surface chemistry, as these elements can play a key role in a robot's interactions with immune system cells.
Unfortunately, designing microrobots with structural parameters that minimize their interactions with the immune system has so far proved to be highly challenging. This is because often, the structural characteristics that tend to activate the immune system are crucial to a robot's functioning and locomotion.
Researchers at Max Planck Institute for Intelligent Systems have recently carried out a study investigating the interplay between structural design parameters of microswimmers for medical applications and the human immune system. Their paper, published in Science Robotics, presents interesting observations that could aid the development of microrobots that do not activate the immune system while retaining a high locomotion performance.
"We investigated the interactions of magnetically steerable double-helical microswimmers with mouse macrophage cell lines and splenocytes, freshly harvested from mouse spleens, by systematically changing their helical morphology," the researchers wrote in their paper.
Essentially, the researchers observed that macrophages and splenocytes, two types of cells with immune functions that they harvested from mice, could recognize helix turn numbers of the microswimmer robots they used, eliciting a targeted immune response. Their findings suggest that the structure of medical microrobots should be optimized taking both their locomotion performance and their interaction with immune cells into close consideration. These two elements should be explored simultaneously in order to find a compromise that allows microswimmers to move efficiently inside the body without interacting with immune cells.
In their paper, the researchers show that morphology-related interactions between macrophages and microrobots could also be leveraged to design new types of biohybrid microrobots. To demonstrate this possibility, they designed 'immunorobots' that merge the steerable mobility of synthetic microswimmers and the immunoregulatory capabilities of immune cells.
In the future, these robots could be used to carry out immunotherapeutic interventions inside human the body. For instance, doctors could steer them close to tumors, in order to limit the impact of immune responses in their vicinity.
More information: Immihan Ceren Yasa et al. Elucidating the interaction dynamics between microswimmer body and immune system for medical microrobots, Science Robotics (2020). DOI: 10.1126/scirobotics.aaz3867
Journal information: Science Robotics
© 2020 Science X Network