February 24, 2014 feature
Self-charging battery gets boost from nanocomposite film
(Phys.org) —In 2012, a research team at the Georgia Institute of Technology led by Professor Zhong Lin Wang fabricated the first self-charging power pack, or battery, that can be charged without being plugged into a wall socket or other source of electricity. Instead, the battery is charged by applying a mechanical stress, which causes lithium ions to migrate from the cathode to the anode due to the piezoelectric effect. Now the researchers have improved the battery by adding nanoparticles to the battery's piezoelectric material, resulting in a higher charging efficiency and storage capacity.
Along with Wang, Yan Zhang and their other coauthors from Lanzhou University, Northeastern University in Shenyang, and the Chinese Academy of Sciences in Beijing (all in China), and the Georgia Institute of Technology, have published a paper on the improved self-charging battery in a recent issue of Nanotechnology.
The self-charging battery is several hundred micrometers thick and fits inside a stainless steel coin-type cell. By placing it underneath the buttons of a calculator, for instance, the mechanical energy generated by pressing a button can be simultaneously converted from mechanical to chemical energy and stored in the battery. The researchers envision that the battery could one day power a variety of small, portable electronic devices.
"Self-charging power cells charged up by mechanical deformation and vibration from the environment have possible applications for self-powered sensor systems, as well as flexible and portable electronics, such as self-charging flexible mobile phones and human health monitoring systems," Zhang told Phys.org.
The self-charging battery's ability to both convert and store energy is what sets it apart from conventional batteries, whose sole purpose is to store energy. In conventional batteries, the first step of energy conversion (such as mechanical to electrical) is almost always performed by a separate device. The self-charging battery completely bypasses the intermediate step of converting to electricity, resulting in a more efficient conversion and storage process than if two separate devices—and two steps—were used.
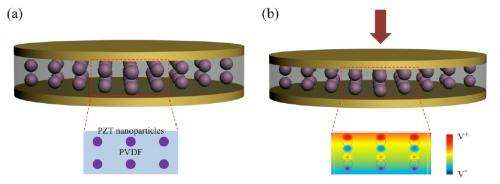
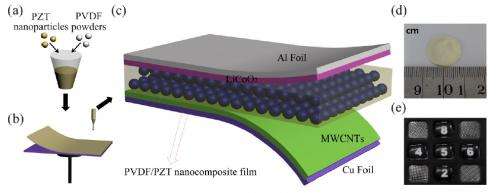
To transform a conventional Li-ion battery into a self-charging one, the researchers replaced the polyethylene separator that normally separates the two electrodes in a Li-ion battery with a piezoelectric material that generates a charge when under an applied stress. In the 2012 version, this material was a PVDF film. In the new study, the researchers added lead zirconate titanate (PZT) nanoparticles to the PVDF film to create a nanocomposite.
The addition of the PZT results in significant performance improvements, namely increasing the battery's efficiency and storage capacity by 2.5 times over the earlier version. Specifically, the storage capacity improved from 0.004 to 0.010 µA h.
The researchers explained that these improvements occur due to two mechanisms: first, the PZT induces a geometrical strain confinement effect that increases the piezoelectric potential; and second, the PZT has a porous structure that increases the number of pores in the nancomposite, resulting in a smaller interpore distance that increases the number of ionic conduction paths on which lithium ions can travel. Both mechanisms allow more lithium ions to migrate from the cathode to the anode, increasing the total charge.
The improvements demonstrate that a nanocomposite film can enhance the performance of self-charging batteries, and the researchers plan to make further improvements in the future.
"We need to deeply understand the exact progress of charging electrochemical reactions at the two electrodes, for improving the performance of the self-charging power cells," Zhang said.
More information: Yan Zhang, et al. "PVDF-PZT nanocomposite film based self-charging power cell." Nanotechnology 25 (2014) 105401 (7pp). DOI: 10.1088/0957-4484/25/10/105401
© 2014 Phys.org. All rights reserved.