Scientists discover dynamics of electrochemical interfaces at the atomic scale

The quest to find viable alternatives to fossil fuels in energy production has experienced a recent revolution as scientists search for materials that do not require precious metals to produce active and stable reactions.
Central to many of these reactions is the oxygen evolution reaction (OER), an important electrochemical part of water-splitting in electrolyzers to produce hydrogen that can power fuel cells.
Scientists at the U.S. Department of Energy's (DOE) Argonne National Laboratory used a combination of high-precision materials science and electrochemistry to provide important insight into the mechanisms that drive stability and activity of materials during the OER. This insight will guide the practical design of materials for electrochemical fuel production.
"Our explanation removes some of the fog surrounding the effects of impurities on stability of a material at both an atomic scale and a macro scale," said Argonne Distinguished Fellow Nenad Markovic, a chemist in the lab's Materials Science division.
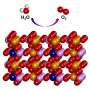
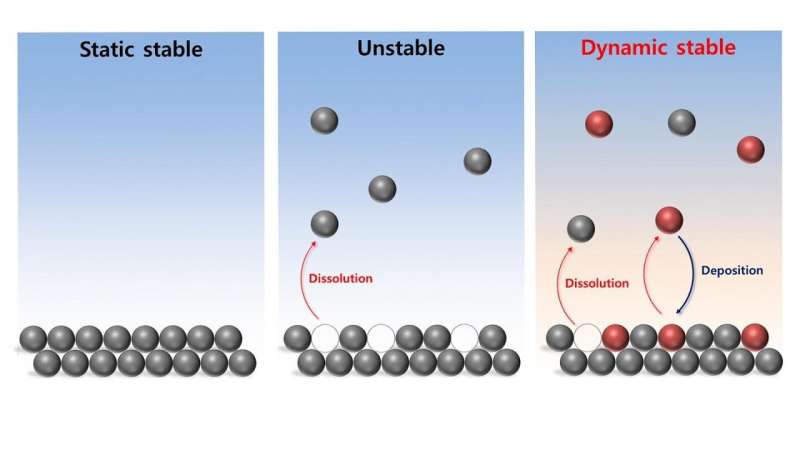
The scientists studied an electrolyzer material, called a hydr(oxy)oxide, to discover that, although electrolyzerscan behave as if they are wholly stable, on an atomic scale the systems are extremely dynamic. Iron atoms present in the electrode repeatedly fall away and reattach to the interface, or the surface on which the important, oxygen-producing reactions take place. This careful balance between dissolution and redeposition allows for the overall stability of the material.

"Traditionally, scientists measure how long an electrolyzer can produce oxygen, and they use that to determine stability," said Argonne postdoctoral scientist Dongyoung Jung, first author on the study. "We decoupled the overall stability of the material on a macro scale from the stability of the material on the atomic scale, which will help us to understand and develop new materials."
The scientists developed ultrasensitive electrochemical measurement tools to monitor the iron activity in situ during the OER and to test the system with various levels of impurities to see what variables affect the overall stability of the material. The behavior of the iron at the interface is responsible for how well the material can produce oxygen in the OER process.
"By measuring the iron content in the electrode and the electrolyte with ultrahigh sensitivity, we found unexpected discrepancies that point to a dynamic stability of the iron in the system," said Pietro Lopes, an Argonne assistant scientist on the study.
The dynamic stability in the material—characterized by stable behavior at the macroscopic level despite high activity at the atomic level—is not necessarily a bad thing for electrolyzers. The scientists hope to take advantage of their new understanding of this phenomenon to create materials with better performance.
"Once we identify the role of iron and how its movement affects the oxygen evolution process, we can modify materials to take advantage of dynamic stability, ensuring that iron is always present at the interface, boosting oxygen production," said Lopes.
"We are addressing a major misconception in the field," said Vojislav Stamenkovic, Energy Conversion and Storage group leader in Argonne's Materials Science division. "The profound implications of the decoupling of virtual stability and true stability will extend the design rules for producing active and stable interfaces."
The study's corresponding paper, published in Nature Energy on March 16, is titled "Dynamic stability of active sites in hydr(oxy)oxides for the oxygen evolution reaction."
More information: Dong Young Chung et al, Dynamic stability of active sites in hydr(oxy)oxides for the oxygen evolution reaction, Nature Energy (2020). DOI: 10.1038/s41560-020-0576-y