This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers design battery prototype with fiber-shaped cathode
In a new study, North Carolina State University researchers made a cathode in the shape of a thread-like fiber. The researchers were then able to use the fiber to create a zinc-ion battery prototype that could power a wrist watch.
Published in the journal ACS Applied Materials and Interfaces, the proof-of-concept study is a step forward in the development of a fiber-shaped battery that could ultimately be integrated into garments.
"Ultimately, we want to make a yarn-shaped battery, so we could put it in a real garment, and preferably hide it," said the study's corresponding author Wei Gao, associate professor of textile engineering, chemistry and science at NC State. "In this study, we created a yarn-shaped cathode. Our findings were pretty promising for such a short strip of fiber, and we hope to continue this work to improve the performance, safety, and mechanical properties of our designs."
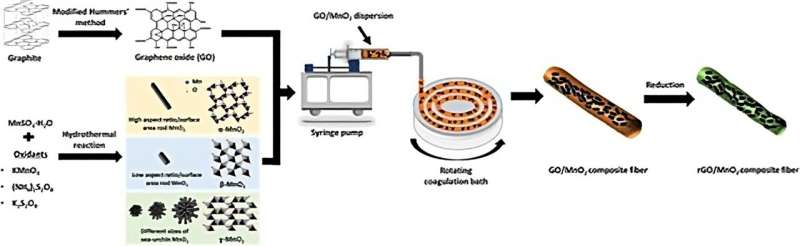
To create the cathode, the researchers utilized the unique properties of graphene—a strong material with a high surface area and good conductivity—to make a yarn-shaped zinc-ion battery.
In their study, the researchers created different manganese dioxide microparticles in various shapes and sizes. Then, they used a solution-spinning process to form a fiber made of graphene oxide, with manganese dioxide particles embedded within. They studied the electrochemical and other properties of the fibers.
"Since we are trying to make a fiber-shaped zinc-ion battery, we care not only about the battery performance, but also about the mechanical properties—we need the fiber to be strong, and also flexible," Gao said.
Researchers found the shape of the graphene oxide and manganese dioxide materials that they used to make the cathode affected its electrochemical function. Specifically, they found the shorter, rod-shaped manganese dioxide components mixed homogeneously with the graphene, allowing researchers to make a fiber that could operate as a functional battery prototype. Comparatively, they found that when the manganese dioxide had a "sea urchin" shape, the fiber couldn't be used in a functional battery.
"When the graphene and manganese dioxide were well-mixed, you can use the fiber to create a functional battery," said the study's first author, Nakarin Subjalearndee, a former graduate student at NC State. "If the manganese dioxide was shaped like a sea urchin, it meant the cathode thread had a rough surface, and could not be used."
"The rod-shaped manganese dioxide gave us the best performance," Gao added. "These particles mimicked the configuration or geometry of the fiber, as compared to the sea urchin-shaped particles, which were round with all kinds of edges pointing out. They disturbed the stacking of the graphene oxide nanosheets within the fiber."
Though the battery's performance was low, the researchers were able to use it to power a wrist watch. The team wants to continue their work to improve the design's performance.
"This study shows that the shape and size of the additives in the fiber affected the fiber formation process of graphene oxide," Gao said. "We hope to keep developing this system; we want our design to be comparable to a commercial battery."
The study, "Wet Spinning of Graphene Oxide Fibers with Different MnO2 Additives," was published online in ACS Applied Materials & Interfaces.
More information: Nakarin Subjalearndee et al, Wet Spinning of Graphene Oxide Fibers with Different MnO2 Additives, ACS Applied Materials & Interfaces (2023). DOI: 10.1021/acsami.3c02989