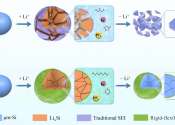
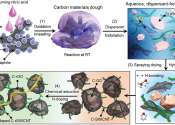
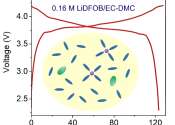
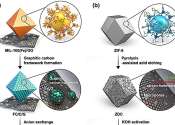
Engineers develop safe and long-cyclable lithium metal battery for high temperatures
In recent years, batteries have become ubiquitous in consumers' daily lives. However, existing commercial battery technologies, which use liquid electrolytes and carbonaceous anodes, have certain drawbacks such as safety ...
Jul 22, 2024
0
47