December 18, 2015 report
Sony pegs 2020 as year of its high capacity battery

(Phys.org)—Instant crowd pleaser: Any headline that promises more battery life for smartphones. And why wouldn't it be a crowd-pleaser, considering the hassles in running out of juice when you need it the most?
Question is, what is the promise and does the claim of better life hold much or any water?
Sony is the latest name in the news making headlines over their work in bringing out a high capacity battery for phones.
Reports from Tokyo such as the one in Nikkei Asian Review said that a new high capacity battery is part of Sony's plans for 2020. The battery, taking up the same amount of space, will mean smartphones can run 40 percent longer—and not only smartphones but other devices too.
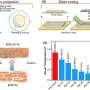
It is no secret that scientists have been searching for a safer, less expensive alternative to lithium-ion batteries. The Joint Center for Energy Storage Research (JCESR) has been working on chemistry, technologies and partnerships that could eventually go beyond lithium-ion batteries.
Problem in brief: "Li-ion cells will never give electric cars the 800-kilometer range of a petrol tank, or supply power-hungry smartphones with many days of juice," said Nature.com last year, in a general discussion about the search for a better battery. That article last year also said that " Li–S (lithium–sulfur technology), batteries were first posited 40 years ago, but researchers could not get them to survive past about 100 cycles. Now, many think that the devices are the technology closest to becoming a commercially viable successor to Li-ion."
An article in ClimateWire in October said that while Lithium-ion batteries can now provide more power for longer durations, consumers want thinner handsets, powerful processors and bigger screens on their phones. The article said that people are still scrounging for power outlets as they were five years ago. As for automobiles, "Electric cars have yet to match the price, performance and range of gasoline engines in this regard. On the power grid, it costs about five times as much to catch and release electricity in a lithium-ion battery as it does to generate fresh power from a gas turbine."
George Crabtree, who leads JCESR, said almost every battery component is a target for innovation.
Meanwhile, the Nikkei Asian Review report said that "Sony's version could power the latest Apple iPhone, the 6s, for 14 hours while the device is connected to the Internet, calculations show."
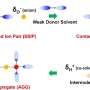
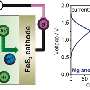
Magic beans? Something more tangible than that, in the form of a sulfur compound. "Batteries used in smartphones and other devices are charged and discharged as ions move from a positive to a negative pole and back again through a liquid electrolyte," said the Nikkei Asian Review report. "Most mainstream batteries use positive electrodes made from lithium cobalt oxide and other such compounds. Sony's battery, however, uses a sulfur compound, letting it store a good deal more electricity than other models."
Sulfur batteries were thought up before Sony did this work but previous attempts at sulfur-bearing batteries saw the electrode dissolve into the electrolyte over repeated charge cycles, shrinking capacity. Sony said it has managed to overcome the problem, in part by reformulating its electrolyte solution.
Battery technology has long been the "turning lead into gold" of the tech industry, said Times of India on Thursday. "The upshot of this technology could be longer lasting batteries but it could also mean smaller batteries - as they will be more efficient. This could mean even thinner phones in the future."
© 2015 Tech Xplore