Could a seawater battery help end our dependence on lithium?
With the ubiquity of lithium-ion batteries in smartphones and other rechargeable devices, it's hard to imagine replacing them. But the rising price of lithium has spurred a search for alternatives. One up-and-coming battery technology uses abundant, readily available seawater. Now, making this option viable is one step closer with a new report on a sodium-air, seawater battery. The study appears in the journal ACS Applied Materials & Interfaces.
Sodium-air—or sodium-oxygen—batteries are considered one of the most promising, and cost-effective alternatives to today's lithium-ion standby. But some challenges remain before they can become a commercial reality. Soo Min Hwang, Youngsik Kim and colleagues have been tackling these challenges, using seawater as the catholyte—an electrolyte and cathode combined.
In batteries, the electrolyte is the component that allows an electrical charge to flow between the cathode and anode. A constant flow of seawater into and out of the battery provides the sodium ions and water responsible for producing a charge. The reactions have been sluggish, however, so the researchers wanted to find a way to speed them up.
For their new battery, the team prepared a catalyst using porous cobalt manganese oxide nanoparticles. The pores create a large surface area for encouraging the electrochemical reactions needed to produce a charge. A hard carbon electrode served as the anode.
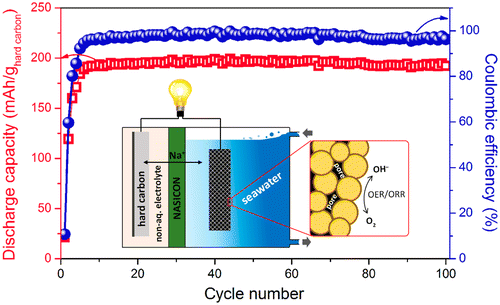
The resulting battery performed efficiently over 100 cycles with an average discharge voltage of about 2.7 volts. This doesn't yet measure up to a lithium-ion cell, which can reach 3.6 to 4.0 volts, but the advance is getting close to bridging the gap, the researchers say.
More information: Mari Abirami et al. A Metal–Organic Framework Derived Porous Cobalt Manganese Oxide Bifunctional Electrocatalyst for Hybrid Na–Air/Seawater Batteries, ACS Applied Materials & Interfaces (2016). DOI: 10.1021/acsami.6b10082
Abstract
Spinel-structured transition metal oxides are promising non-precious-metal electrocatalysts for oxygen electrocatalysis in rechargeable metal–air batteries. We applied porous cobalt manganese oxide (CMO) nanocubes as the cathode electrocatalyst in rechargeable seawater batteries, which are a hybrid-type Na–air battery with an open-structured cathode and a seawater catholyte. The porous CMO nanocubes were synthesized by the pyrolysis of a Prussian blue analogue, Mn3[Co(CN)6]2·nH2O, during air-annealing, which generated numerous pores between the final spinel-type CMO nanoparticles. The porous CMO electrocatalyst improved the redox reactions, such as the oxygen evolution/reduction reactions, at the cathode in the seawater batteries. The battery that used CMO displayed a voltage gap of ∼0.53 V, relatively small compared to that of the batteries employing commercial Pt/C (∼0.64 V) and Ir/C (∼0.73 V) nanoparticles and without any catalyst (∼1.05 V) at the initial cycle. This improved performance was due to the large surface area (catalytically active sites) and the high oxidation states of the randomly distributed Co and Mn cations in the CMO. Using a hard carbon anode, the Na-metal-free seawater battery exhibited a good cycle performance with an average discharge voltage of ∼2.7 V and a discharge capacity of ∼190 mAh g–1hard carbon during 100 cycles (energy efficiencies of 74–79%).