Study of interface thickness evolution on high-capacity battery electrodes
Wouldn't it be nice if lithium-ion batteries lasted as long as a car? Scientists have proposed silicon as a high-capacity negative electrode for lithium-ion batteries. Unfortunately, the formation and thickening of interface layers on top of silicon electrodes degrades cell performance. The result? You have to replace the battery. Measurements of the thickness of the interface material, known as the solid-electrolyte interphase or SEI, vary by two orders of magnitude. In this research, scientists combined real-time atomic force microscopy, which can measure surface layer thicknesses, with electrochemical cycling and a carefully designed sample geometry to unambiguously measure the SEI evolve. The sample design facilitates clear separation of SEI thickness evolution from the volume changes of the underlying silicon electrode. The measurements can be used to benchmark models of SEI reaction kinetics for different electrolytes.
To lengthen the life of lithium-ion batteries, we need to know how to combat SEI formation. This measurement capability is a vital step in new studies of SEI properties. The approach offers real-time measurement of SEI thickness evolution with nanometer precision. It will allow for effective study of electrolyte compositions and additives and their effect on cell performance.
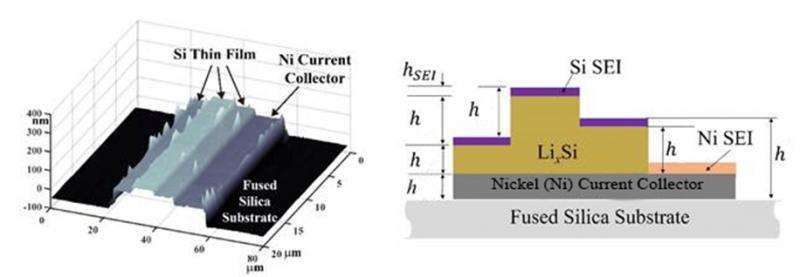
This research has developed new techniques for in situ measurements of the growth of solid-electrolyte interphase (SEI) layers on silicon electrodes. These interface changes are accompanied by volume expansion induced by transport of lithium into the silicon. For these investigations, the team fabricated thin film amorphous silicon electrodes in a configuration that allows unambiguous separation of the total thickness change into the contributions due to the growth of the SEI and from the change in the silicon volume.
They assembled the electrodes into a custom-designed electrochemical cell, which they integrated with an atomic force microscope. The electrodes are subjected to charge-discharge (lithiation-delithiation) cycles at a sequence of constant potential values and the thickness measurements are made at each potential after equilibrium is reached. The team carried out experiments with two electrolytes: 1.2M lithium hexafluoro-phosphate (LiPF6) in ethylene carbonate (EC) and 1.2M LiPF6 in propylene carbonate (PC) – to investigate the influence of electrolyte composition on SEI evolution.
The team observed that SEI formation occurs predominantly during the first charging cycle and the maximum SEI thickness is approximately 17 nm and 10 nm, respectively, for EC and PC electrolytes. The measurements also yield valuable information on how the silicon electrode's expansion ratio and charge capacity vary with equilibrium potential.
Both relationships display hysteresis (for example, the measurements show a dependence on whether the electrode is being charged or discharged), which is explained in terms of the evolution of stress in silicon electrodes due to the changing volume. This sample design and measurement capability open the door for clear determination of which components and variations result in performance improvements in high-capacity lithium-ion batteries.
More information: Insun Yoon et al. In Situ Measurement of Solid Electrolyte Interphase Evolution on Silicon Anodes Using Atomic Force Microscopy, Advanced Energy Materials (2016). DOI: 10.1002/aenm.201600099