April 19, 2019 report
Adding guanidinium thiocyanate to mixed tin-lead perovskites to improve solar cell efficiency

A team of researchers affiliated with several institutions in the U.S. has found a way to improve the efficiency of perovskite-based solar cells—by adding guanidinium thiocyanate to the mix. In their paper published in the journal Science, the group describes their work with perovskite-based solar cells and how well they worked.
For most of its history, silicon has been the material of choice when making solar cells—no other material was as efficient or could produce for as long. But in recent years, chemists have been working with different materials that have come closer. One such promising material is crystalline perovskite. It is generally made from lead, bromine, iodine and other elements. Currently, solar cells made with perovskite have two advantages over traditional silicon cells. They are cheaper to make and they are better at absorbing high-energy blue photons. The second feature has led solar cell makers to marry the two types of cells together to create tandem silicon/perovskite cells providing the best benefits of both. But solar cell makers still believe that eventually, all-perovskite-based cells can replace silicon cells entirely at some point, resulting in bringing down production costs. In this new effort, the researchers claim to have found a way to come close.
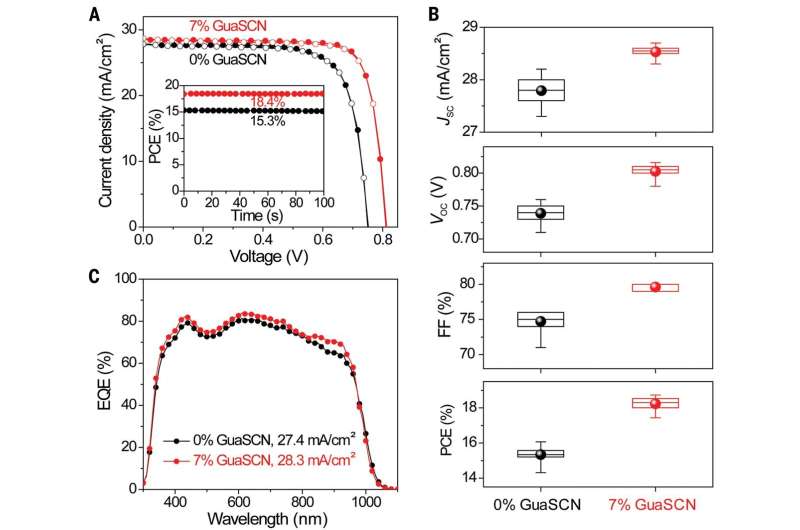
Prior research had shown that adding tin to the mix when making perovskites resulted in making them more efficient—almost as efficient as silicon-based cells. But tin degraded when exposed to oxygen. To prevent that from happening, the team in the U.S. also added guanidinium thiocyanate to the mix. It is an organic compound that coats other materials—in this case, the tin in the perovskite mix. The researchers found that doing so prevented the tin from degrading. Testing the resulting perovskite showed it to be approximately 20 percent efficient. When the team combined it with a traditional perovskite cell designed to absorb high energy photons—making an all-perovskite tandem—they saw efficiencies of 25 percent. This is close to the 28 percent seen with silicon-perovskite tandems.
The researchers note that they believe they can increase the efficiency more, perhaps reaching silicon/perovskite tandem levels, but acknowledge they still have other issues to deal with before such cells would become viable—most prominently, making them last long enough for commercial use.
More information: Jinhui Tong et al. Carrier lifetimes of >1 μs in Sn-Pb perovskites enable efficient all-perovskite tandem solar cells, Science (2019). DOI: 10.1126/science.aav7911
© 2019 Science X Network