July 17, 2019 feature
Inspired by water lilies: A hierarchical design for solar evaporation of high salinity brine
Interfacial solar vapor generation has great potential for desalination and wastewater treatment with high energy conversion efficacy. High water evaporation rates cannot be maintained using existing techniques, however, due to unavoidable fouling or salt accumulation on the solar absorbers that cause accelerated degradation of the devices. In a recent study, Ning Xu and colleagues at the National Laboratory of Solid State Microstructures, College of Engineering and Applied Sciences and Artificial Functional Materials in China have demonstrated a water lily-inspired hierarchical structure to facilitate efficient solar evaporation of high salinity brine and wastewater.
The experimental device allowed the evaporation of high-salinity brine and wastewater containing heavy metal ions, without decreasing evaporation rates or fouling the absorbers during the entire process to completely separate the water and the solute. The new and improved method will have direct implications in a variety of fields such as wastewater treatment facilities as well as sea-salt production and metal recycling. The results of the study are now published on Science Advances.
Freshwater scarcity is a serious global crisis due to increasing global human population and substantial levels of consumption and contamination of freshwater bodies. Scientists developed a variety of water treatment technologies, including reverse osmosis (RO) and ultrafiltration to ease pressure from continued freshwater withdrawal and reduce the environmental impact of discharged wastewater. For instance, Zero Liquid Discharge (ZLD) was proposed as an ultimate technique to completely separate solute and water for reuse, while minimizing harmful ecological impacts during wastewater treatment. However, an energy efficient and cost-effective method to treat concentrated brine remains to be developed for large-scale ZLD implementations.
A significant challenge with mainstream techniques such as RO is the dramatic pressure (energy) required during the filtering process with increased brine concentration; resulting in high energy costs for water treatment. As a result, new pathways should be explored with specific focus on high-concentration brine or wastewater to completely separate water and the solute with minimal energy costs coupled to long-term environmental safety. Researchers had previously explored several strategies to maintain high evaporation rates from concentrated brine across a long period of time. Examples include Mangrove tree inspired "artificial leaves," methods to increase the water supply to dissolve salt sediments on absorbers and solar desalination with relatively stable performances for 120 hours.
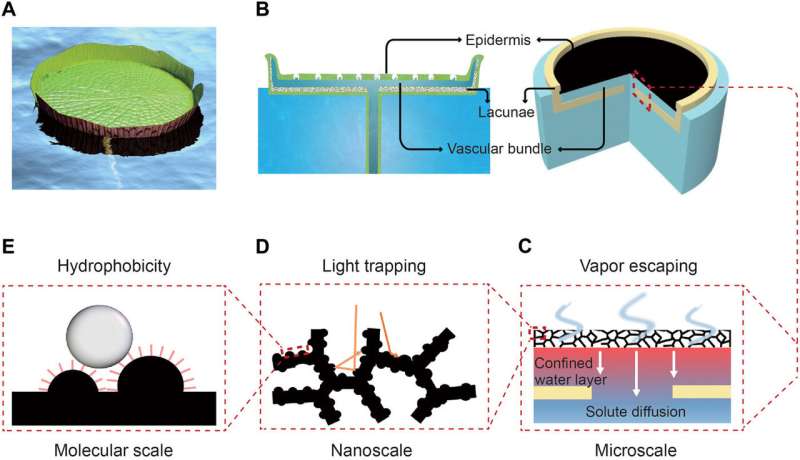
A water lily-inspired design introduced in the present work is an elegant system of transpiration with several native features. In a natural water lily, the primary feature is an upper epidermis that absorbs sunlight in the company of stomata for water vapor escape while retaining a hydrophobic self-cleaning surface. As a second feature, the plant can naturally float on the surface of water due to an air chamber (lacunae) that exists at the bottom of the leaf. Third, the floral design can be confined to a water path that pumps up water through vascular bundles and spread them to the surface of the structure. Xu et al. mimicked these features to propose a water lily-inspired hierarchical structure (WHS) and realize highly efficient and stable solar evaporation in high-salinity brine/wastewater for complete water and solute separation.
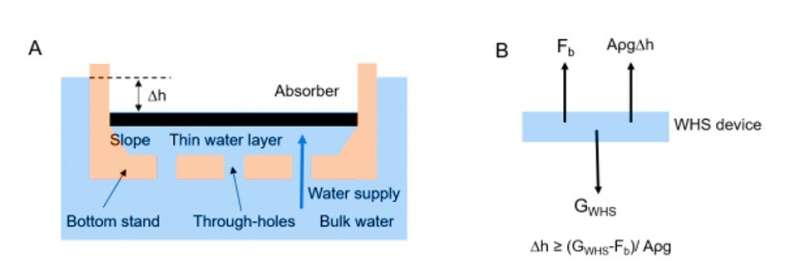
In the new WHS device, Xu et al. mimicked the water lily, beginning with a top solar absorber hierarchically designed to absorb sunlight and provide continuous vapor escape through "artificial stomata." They developed nanostructure surface modifications on the solar absorber for hydrophobic properties—much like the water lily; preventing water entry into the absorber for efficient solar evaporation. Similar to the lacunae (air chamber) of the plant, a bottom stand supported the entire structure to naturally float on water while serving as a thermal insulation layer to minimize heat losses. Like its natural counterpart, the WHS only supported water to rise up through the confined channels containing holes in the bottom stand.
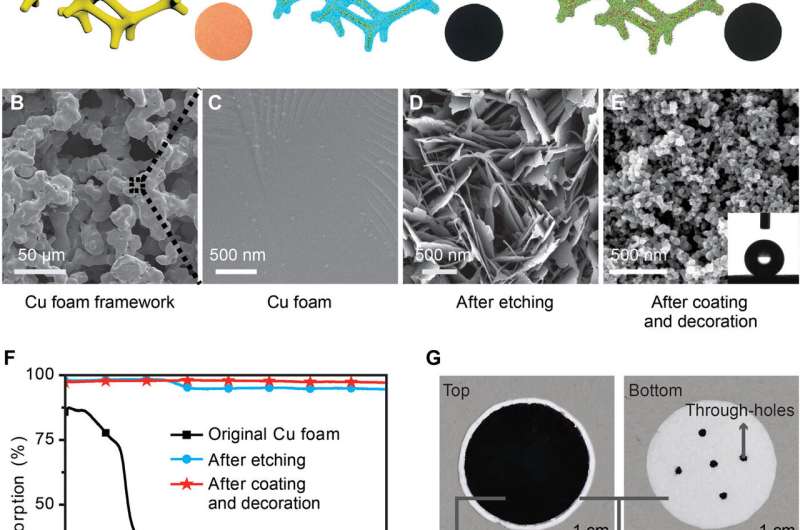
The scientists chose copper (Cu) foam as the initial substrate to develop the WHS due to its high thermal conductivity and micron-sized pores to facilitate vapor escape. They then transformed the smooth surface of Cu into knife-like nanoplates using chemical etching to engineer nanoscale light trapping effects and enhance solar absorption. They followed this step by coating the surface absorber with a layer of Aluminum oxide (Al2O3) decorated with carbon black (CB) nanoparticles to protect the surface and enhance sunlight absorption at infrared (IR).
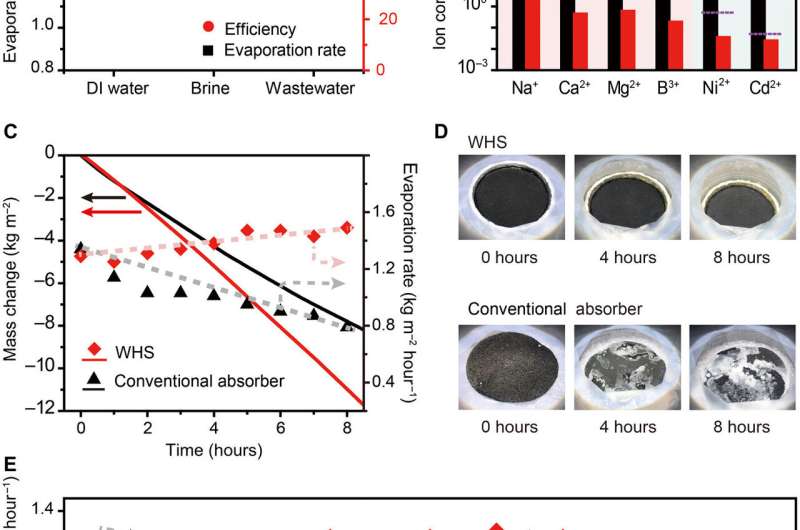
To test the evaporation performance of WHS, Xu et al. treated deionized water, 10 percent weight brine and 30 percent weight wastewater (containing a heavy metal solution) under one-sun illumination. When they monitored the evaporation rates, the values were comparable to high-performance solar absorbers as reported previously. When they tested the purification effect of desalination and wastewater treatment via WHS, the ion concentrations in seawater (Na+, Mg2+, Ca2+) and wastewater (Ni2+, Cd2+) or Na+ in brine were significantly reduced. The purification standards met the World Health Organization (WHO) standard for drinking water or standard for discharge.
To test long-term stability of the device, Xu et al. conducted a continuous, eight-hour experiment of solar water treatment under a solar simulator in the lab to account for the absence of performance degradation and fouling issues. For this, they compared the WHS and a conventional solar absorber with similar evaporation rates demonstrated for pure water. During the desalination experiments, the surface of the WHS remained clean throughout evaporation to indicate its antifouling capability.
Comparatively, salt progressively accumulated on the conventional solar absorber surface, blocking sunlight absorption (energy input). Xu et al. noted the average evaporation rate of WHS to be much higher than that of the solar absorber during 8 hours of the experiment. When they performed a similar experiment across 18 days outdoors under natural sunlight, the evaporation rate was stable for WHS and decreased for conventional solar absorbers.
When treating brine and wastewater under solar absorption, the WHS notably facilitated the complete separation of water and the solute. Thereafter, the scientists easily removed the remaining salt/solute crystals after complete water evaporation. In this way, Ning Xu and co-workers demonstrated a new WHS device that can conduct fast and stable evaporation across long-term treatment of high-salinity brine or highly concentrated saltwater. They achieved complete separation of the water and solute without fouling (salt/solute accumulation) on the device. The scientists expect the device to have direct implications in a variety of applications including sea-salt production, resource recovery and chemical fractionation in the near future.
More information: Ning Xu et al. A water lily–inspired hierarchical design for stable and efficient solar evaporation of high-salinity brine, Science Advances (2019). DOI: 10.1126/sciadv.aaw7013
R GOPAL et al. Electrospun nanofibrous filtration membrane, Journal of Membrane Science (2006). DOI: 10.1016/j.memsci.2006.04.026
M. Elimelech et al. The Future of Seawater Desalination: Energy, Technology, and the Environment, Science (2011). DOI: 10.1126/science.1200488
Pedro J. J. Alvarez et al. Emerging opportunities for nanotechnology to enhance water security, Nature Nanotechnology (2018). DOI: 10.1038/s41565-018-0203-2
© 2019 Science X Network

















