February 17, 2020 feature
An approach to fabricate stable perovskite quantum dot solar cells with high power conversion efficiencies
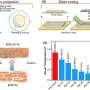
![Morphology and crystal structure of Cs1−xFAxPbI3 QD. a,b, Low-magnification (a) and high-magnification (b) HAADF–STEM images of Cs0.5FA0.5PbI3 QDs. c,d, Representative atomic-resolution HAADF–STEM image (c) and the derived colour-coded HAADF-STEM image (d) of Cs0.5FA0.5PbI3 QD. The image is viewed along the cubic [001] zone axis established from the lattice arrangement similar to the original CsPbI3 (or FAPbI3) cubic crystal structure. (e–g) The enlarged view (e), the atomic models (f) and the simulated atomic-resolution HAADF–STEM image (g) for the region marked with a red square in c and d. In e, the yellow dots represent the atom column in the area with uniform contrast distribution; the pink and blue dots (pink dots represent the high-contrast atoms and blue dots represent the low-contrast atoms) represent the atom columns in the area with distinct contrast distribution. All the experimental STEM images of the samples containing FA are distorted due to decomposition of the local crystal structure from FAPbI3 to PbI2 under electron beam illumination. h, The simulated atomic-resolution HAADF–STEM image. i, The atomic models for the original view of Cs0.5FA0.5PbI3 QD along the [001] zone axis after reconstruction. j, The established crystal structure of the Cs1−xFAxPbI3 QD. Scale bar, 10 Å (e,g,h). Credit: Hao et al. An approach to fabricate stable perovskite quantum dot solar cells with high power conversion efficiencies](https://scx1.b-cdn.net/csz/news/800a/2020/anapproachto.jpg)
Past research studies have highlighted the potential of perovskite materials for the development of several technological tools, including photovoltaics (PVs) and optoelectronics. Solution-processed organic-inorganic lead halide perovskite materials have been found to be particularly promising, especially those with a common ABX formulation, where A is an organic cation, B is lead (Pb) or tin (Sn) and X is a halide.
These materials have several advantageous optoelectronic properties, including a large absorption coefficient, a long carrier diffusion length and a low exciton dissociation energy. Solar cells made of these materials have recently been found to achieve power conversion efficiencies (PCEs) that match or exceed those of more conventional solar cells made of silicon, cadmium telluride and copper indium gallium selenide.
Despite their advantages, solar cells made of perovskites with a common ABX formulation can have a number of limitations, including fast degradation. One of the most promising perovskite-based compositions in terms of stability, CS1-xFAxPbI3, has also been found to result in solar cells presenting large open-circuit voltage losses, which has so far prevented it from being implemented on a larger scale.
Researchers at the University of Queensland, Swansea University and other institutions worldwide have recently proposed a new strategy that could enable the creation of more reliable solar cells made of CS1-xFAxPbI3, helping to overcome some of the shortcomings reported in past studies. This strategy, presented in a paper published in Nature Energy, allows for the controllable synthesis of CS1-xFAxPbI3 materials, which has so far proved very challenging.
"The mixed caesium and formamidinium lead triiodide perovskite system (CS1-xFAxPbI3) in the form of quantum dots (QDs) offers a pathway toward stable perovskite-based photovoltaics and optoelectronics," the researchers wrote in their paper. "However, it remains challenging to synthesize such multinary QDs with desirable properties for high-performance QD solar cells (QDSCs)."
The researchers essentially proposed a strategy that can be used to synthesize mixed-cation CS1-xFAxPbI3 materials and control some of their properties so that they can be used to fabricate solar cells with a high performance and stability. In their experiments, this strategy allowed them to identify a particular version of the material, namely Cs0.5FA0.5PbI3 witha remarkable PCE of 16.6% and negligible hysteresis.
The team used this material to create QD devices and conducted a series of tests evaluating their performance. Remarkably, the devices presented a photostability comparable to that of thin-film materials, retaining 94% of their original PCE under continuous one-sun illumination over a period of 600 hours.
"We report an effective oleic acid (OA) ligand-assisted cation-exchange strategy that allows the controllable synthesis of CS1-xFAxPbI3 QDs across the whole composition range (x=0-1), which is inaccessible in large-grain polycrystalline thin films," the researchers wrote in their paper. "In an OA-rich environment, the cross-exchange of cations is facilitated, enabling the rapid formation of CS1-xFAxPbI3 QDs with reduced defect density."
In the future, the OA ligand-assisted cation-exchange strategy devised by this team of researchers could pave the way toward the fabrication of new perovskite-based solar cells and optoelectronic technologies that are highly efficient, while also exhibiting high photostability and flexibility. Their work could also inspire other researchers to devise similar strategies, enabling the synthesis of other solution-processed organic-inorganic perovskite materials.
More information: Mengmeng Hao et al. Ligand-assisted cation-exchange engineering for high-efficiency colloidal Cs1−xFAxPbI3 quantum dot solar cells with reduced phase segregation, Nature Energy (2020). DOI: 10.1038/s41560-019-0535-7
© 2020 Science X Network