First millimeter-scale wireless transceiver for electronic pills

This week, at ISSCC 2020 (Feb 16-20, San Francisco), imec, a world-leading research and innovation hub in nanoelectronics and digital technologies, presents the first mm-scale wireless transceiver for smart insertable pills. It is a first breakthrough in imec's aspiration to realize autonomous ingestible sensors that can measure health parameters such as gut health and transmit in real time the data outside the body.
Digestive processes and gastrointestinal diseases are hard to diagnose. Current procedures like endoscopic inspection and stool sample analysis are uncomfortable and only provide one-time observations. Ingestible health trackers, small enough to be swallowed, could collect information over a longer period of time and transmit the data outside the body. Moreover, fixating the ingestible sensors along the gastrointestinal tract, enables longer recordings in specific places of interest. Also, this would increase the comfort of the patient, as he or she could stay out of the hospital while data are collected and sent in real time to the doctor.
Up to 30 times smaller volume than the state-of-the-art
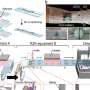
One of the challenges in realizing electronic pills is developing a wireless link that meets the volume, power and performance constraints for reliable data transmission during the period of time that the sensor is collecting data inside the body. Imec's new wireless transceiver supports the medical 400 MHz frequency bands such as MICS (Medical Implant Communication Service), MEDS (Medical Data Service) or MedRadio (Medical Device Radiocommunications Service). The wireless transceiver is implemented in 40 nm CMOS and includes on-chip tunable matching network (TMN) that enables a miniature 400 MHz antenna, and as such avoids external and bulky matching components. The whole transceiver module including antenna occupies a volume of < 55 mm3, which is up to 30 times smaller than state-of-the-art devices. The whole wireless module occupies an area of 3.5 by 15 mm2, including a 3.5 by 3.8 mm2 PCB and a miniature 400MHz antenna. The small form factor is realized thanks to a new crystal-free transceiver architecture, alleviating the need for an off-chip crystal device, and a 2 mm2 transceiver IC with the on-chip TMN. Small area is further achieved by a TX/RX shared matching network with only one on-chip inductor and a single-branch phase tracking RX.
Reliable data transmission outside the body
While inside the body the pills are covered my multiple layers of tissue. Therefore, a wireless system operating in the 400MHz medical bands is preferable because of lower tissue attenuation compared to higher-frequency bands and ensures a reliable and interference-free channel for the implants. Furthermore, the on-chip tunable matching network using self-calibration expands impedance matching up to a VSWR (voltage standing wave ratio) of 4.8, covering a wide range of impedance variation in the body, equivalent to a filled and empty stomach. To realize crystal-free operation, imec implemented an over-the-air network-assisted carrier recovery for large frequency offsets of up to 320 ppm.
"Smart ingestible pills and smart implants offer endless possibilities in what can be measured and treated in the body. This trend leverages on the miniaturization revolution in nanoelectronics, that enables smart, small and lightweight devices with minimal power consumption and maximal patient comfort. However, the development of such devices brings along a specific set of challenges," stated Chris Van Hoof, vice president connected health at imec and general manager of OnePlanet research center. "Through our longstanding leadership in microchip technology and in-depth expertise in software and ICT, we are in the excellent position to develop the necessary building blocks for smart medical implants, related to sensing, actuation, powering, wireless communication, biocompatibility and data processing. Our R&D activities range from early research developments to clinical validation of prototypes."