May 26, 2020 feature
A method to produce hydrogen in vivo photosynthetically
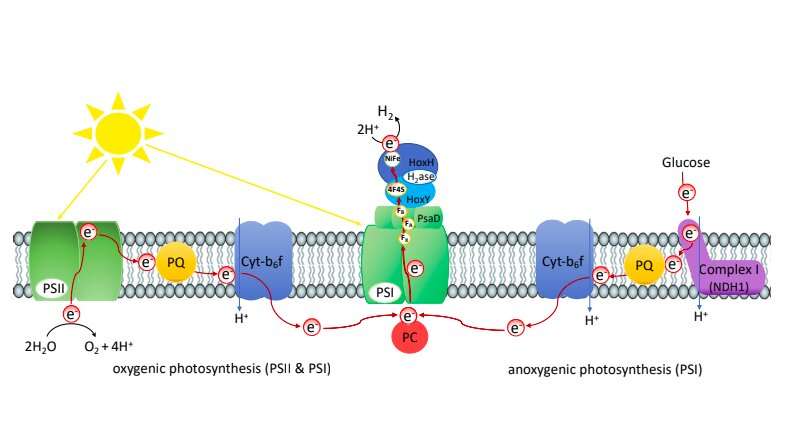
Researchers have been trying to produce hydrogen photosynthetically for quite some time now, as this could pave the way toward a more sustainable energy infrastructure. Some of them have succeeded by fusing different hydrogenases (H2ase) to photosystem I (PSI) in vitro outside of living cells. Hydrogenases are enzymes that catalyze the reduction of protons to hydrogen, while photosystem I is an integral membrane protein complex, which typically captures the energy of sunlight in the process of photosynthesis.
Past studies introduced a number of techniques to fuse hydrogenases to photosystem I, but all of these have so far only proved effective in vitro (i.e., in laboratory settings and outside of living cells). In vitro systems have an intrinsically short lifespan, as the living cell that is capable of self-repair, maintenance and replication is missing. To be most effective, therefore, techniques for the production of solar hydrogen should also work well in vivo (i.e., inside living cells).
Researchers at University Kiel in Germany have recently devised a method to photosynthetically produce hydrogen that entails fusing hydrogenase and photosystem I in vivo. Their paper, published in Nature Energy, could open up new possibilities for the sustainable production of hydrogen on a large scale.
"One challenge for in vivo approaches is that the cells naturally need electrons coming from photosystem I for CO2 fixation via the Calvin-Benson cycle," Kirstin Gutekunst, who led the research group, told TechXplore. "They are obviously not interested in redirecting these electrons to a hydrogenase for hydrogen production."
The recent study carried out by Gutekunst and her colleagues had two key objectives. First, the researchers wanted to combine a hydrogenase and photosystem I in a way that enhanced the production of hydrogen, as compared to its production in wildtype cells (i.e., in their naturally occurring form). In addition, they wanted to ensure that the cells they mutated could grow photoautotrophically, retaining their ability to fix CO2.
"Luckily, structures of photosystem I are around and the electron transfer through this complex is known in detail," Gutekunst said. "Several FeS clusters in photosystem I are involved in transferring electrons to its surface. In the wildtype, these electrons are accepted from a ferredoxin which eventually provides them to the Calvin-Benson cycle for CO2-fixation. The hydrogenase does as well contain FeS clusters that accept electrons and transfer them to its active site for hydrogen production."
Past research has consistently found that to enable an effective transfer of electrons, distances between FeS clusters should be within a specific range. Gutekunst and her colleagues thus tried to fuse the hydrogenase and photosystem I in a way that ensured that distances between FeS clusters of the photosystem and the hydrogenase were fairly small, ultimately enabling a steady photosynthetic electron flux toward the hydrogenase for hydrogen production.
"There were a lot of considerations involved in our design," Gutekunst explained. "However, in the end, we were lucky as well that our system is leaky enough to allow the cells to still fix CO2 and that they also produce photosynthetic hydrogen."
In the experiments carried out by Gutekunst and her colleagues, their solar hydrogen production method led to the creation of a psaD-hoxYH mutant that still grows photoautotrophically. This enabled the photosynthetic production of a high concentration of hydrogen, more specifically 500 μM in the light, under anaerobic conditions.
The researchers are among the first to introduce a proof of concept for the efficient production of solar hydrogen through the fusion of photosystem I and hydrogenase in vivo. Before their method can be implemented in real-world settings, however, they will need to optimize it and overcome a few technical challenges.
"The next steps will be to optimize the electron transfer from photosystem I to the hydrogenase by testing new fusions in which the distances of the FeS clusters of photosystem I and the hydrogenase are smaller," Gutekunst said. "Another very important task is to base the hydrogen production exclusively on oxygenic photosynthesis and water splitting at photosystem II."
So far, the production of hydrogen in the mutant cells attained by Gutekunst and her colleagues is achieved through both oxygenic and anoxygenic photosynthesis. This means that the electrons that enable the production of hydrogen in their system are derived in part from the splitting of water and in part from other sources (e.g., glucose).
Furthermore, the utilized hydrogenase is oxygen sensitive, which poses a challenge for their system's implementation as water splitting at photosystem II results in the evolution of oxygen that inactivates its enzyme. One approach could be to utilize oxygen tolerant hydrogenases from other organisms.
"In our study, we fused the hydrogenase to photosystem I in the cyanobacterium Synechocystis sp. PCC 6803," Gutekunst added. "I find it exciting that more or less at the same time a research group led by Kevin Redding successfully fused the hydrogenase to photosystem I in the green algae Chlamydomonas reinhardtii. We thus now have two existing in vivo systems that produce solar hydrogen in vivo by means of photosystem I—hydrogenase fusions."
More information: Jens Appel et al. Cyanobacterial in vivo solar hydrogen production using a photosystem I–hydrogenase (PsaD-HoxYH) fusion complex, Nature Energy (2020). DOI: 10.1038/s41560-020-0609-6
© 2020 Science X Network



















