June 11, 2020 report
Drug-carrying platelets engineered to propel themselves through biofluids
A team of researchers from the University of California San Diego and the University of Science and Technology Beijing has developed a way to engineer platelets to propel themselves through biofluids as a means of delivering drugs to targeted parts of the body. In their paper published in the journal Science Robotics, the group outlines their method and how well it worked when tested in the lab. In the same issue, Jinjun Shi with Brigham and Women's Hospital has published a Focus piece outlining ongoing research into the development of natural drug delivery systems and the method used in this new effort.
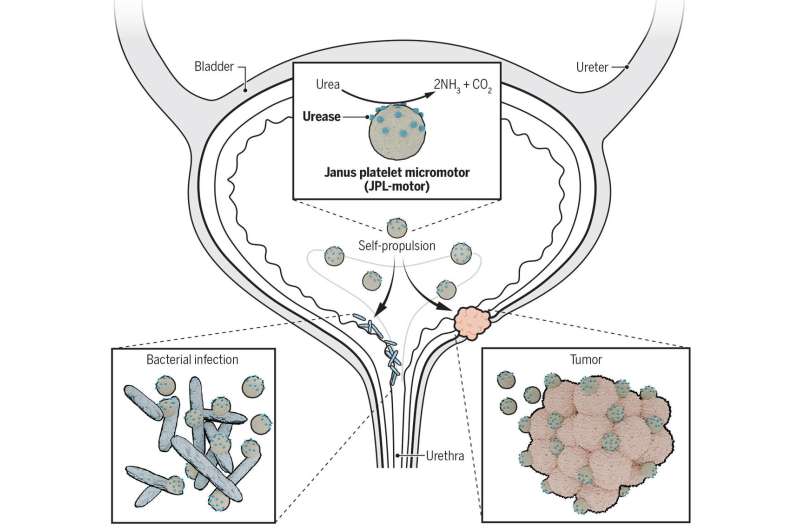
Medical scientists have been working with roboticists over the past several years to determine if it might be possible to launch tiny robots into the human body to carry drugs to specific parts of the body, such as an organ with a bacterial infection or a cancerous tumor. Most such efforts have involved injecting tiny capsules with metallic coatings that can be controlled using an external magnet. But as Shi notes, such efforts tend to be quite inefficient. Because of that, researchers have started to look at the possibility of engineering natural cells in the body to perform as programmed robots. In this new effort, they have devised a way to allow platelets to propel themselves through biofluids. Platelets, Shi also notes, were a good candidate because they are naturally able to carry material around in the body.
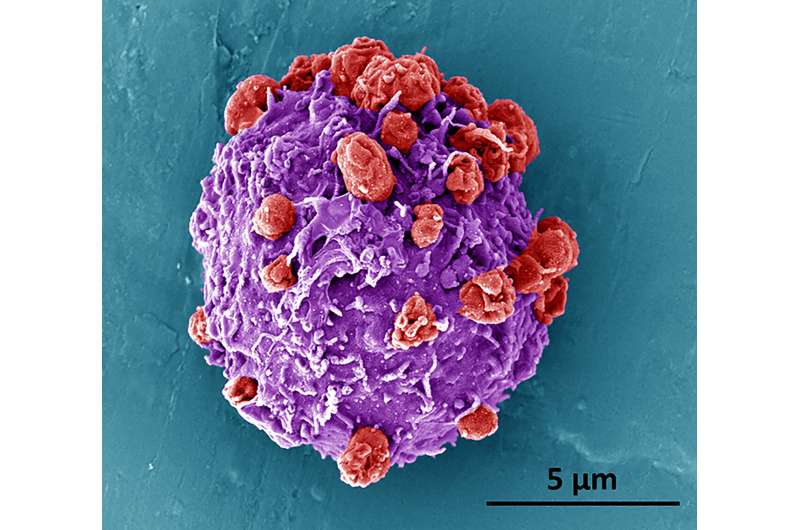
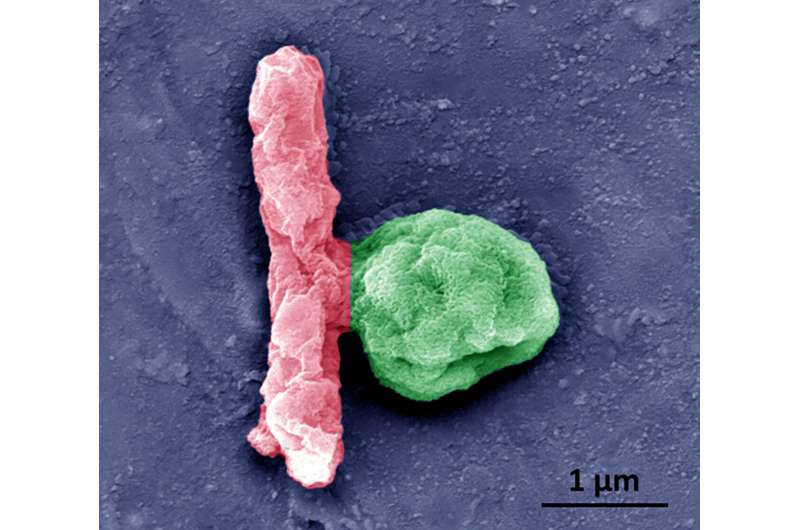
Under normal conditions, platelets are not able to move on their own; they are transported through the blood to different parts of the body. To give them a means of propulsion, the researchers asymmetrically coated them with an enzyme called urease—when it is exposed to urea, a reaction occurs that results in a force that can be used to propel the platelet. By coating the platelets asymmetrically, the team ensured that they were pushed in just one direction. The researchers noted that the speed of the platelet movement could be controlled by the concentration of the urease—and that the application of urease did not harm the platelet surface or its protein profile.
More information: Songsong Tang et al. Enzyme-powered Janus platelet cell robots for active and targeted drug delivery, Science Robotics (2020). DOI: 10.1126/scirobotics.aba6137
© 2020 Science X Network