June 2, 2020 feature
New electrocatalysts to produce ethanol more efficiently

In recent years, researchers worldwide have been exploring new methods of producing ethanol, a chemical compound that is extensively used in a variety of industrial settings. One way to produce this compound is via what is known as the carbon dioxide electroreduction reaction (CO2RR).
While this particular reaction has been found to enable the conversion of CO2 into ethanol, the Faradaic efficiency and current efficiency of the resulting ethanol is often far from optimal. The Faradaic efficiency is essentially the charge efficiency with which electrons are transferred, and utilized to synthesize the desired chemical product, ethanol in this case.
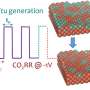
Researchers at University of Toronto have recently devised a strategy that enables a more efficient conversion of CO2 into ethanol via the suppression of deoxygenation in the CO2RR reaction. In their paper, published in Nature Energy, they propose the use of a specific class of catalysts that lead to the production of ethanol with a Faradaic efficiency of (52 ± 1)% and a cathodic energy efficiency of 31%.
"The goal of our project was to boost both the selectivity towards ethanol and its production rate, all under the CO2RR reaction," Dr. Xue Wang, one of researchers in Prof. Ted Sargent's group at University of Toronto who carried out the study, told TechXplore. "To determine how to select in favor of ethanol, we needed to first consider the branching between ethanol and ethylene, its main competing product."
Ethanol and ethylene share an intermediate derivative compound, called HOCCH*. The key element that favors the production of ethanol over that of ethylene via the CO2RR reaction is the suppression of the C-O bond breaking in HOCCH*.
Prof. Sargent and his colleagues designed a set of catalysts by overcoating Cu with nitrogen-doped carbon (N-C/Cu), via a process known as sputter deposition. The catalysts they designed exploit the fact that supressing deoxygenation in HOCCH* promotes ethanol rather than ethylene production.
"Electron microscopy revealed gaps having width typically less than 1 nm between Cu and N-C layers; these serve as nanoreactors in CO2RR," Prof. Ted Sargent told TechXplore. "With the aid of calculations and simulations, as well as in-situ Raman and X-ray absorption, we found that the confinement of the N-C layer offers strong electron-donating ability atop the Cu catalyst and facilitates C-C coupling while suppressing the deoxygenation of the intermediate HOCCH* to ethylene."
The unique properties of the N-C/Cu catalysts devised by Prof. Sargent, Dr. Wang, and their colleagues promote selectivity to ethanol in a more efficient way. Using these catalysts, the researchers achieved CO2-to-ethanol conversion with a record Faradaic efficiency of 52% at a partial current density of 156 mA cm-2.
"The corresponding cathodic energy efficiency (EE) and full cell EE for ethanol reached values of 31% and 16% in flow cell and membrane electrode assembly systems, respectively," Dr. Wang said. "Our study thus provides a new platform to improve the high-energy-density ethanol selectivity at high production rates in CO2RR."
The new catalysts introduced by this group of researchers could enable the more efficient production of ethanol on a large scale. The renewable ethanol made utilizing the Toronto-based team's electrolyzer approach has the potential to displace existing techniques to produce ethanol, which result in compounds with a larger carbon footprint.
The largest application of ethanol today is as engine fuel and as a fuel additive in internal combustion engines. For example, in the United States, both E10 (10% ethanol, sometimes known as gasohol) and E85 (85% ethanol) ethanol/gasoline mixtures are widely sold. As a result, the global ethanol market exceeds $30B USD/year today.
"While this work is a significant step in the right direction, further progress is needed," Prof. Sargent said. "Further research in this filed will include the further improvement of selectivity, production rates, operational stability and energy-efficiency (EE), for all ethanol produced via CO2RR. Further progress efficiency remains our leading priority."
More information: Xue Wang et al. Efficient electrically powered CO2-to-ethanol via suppression of deoxygenation, Nature Energy (2020). DOI: 10.1038/s41560-020-0607-8
© 2020 Science X Network