Carbon dioxide removal from the atmosphere using sustainable energy

Much work is taking place on methods for capturing CO2 from the atmosphere to combat climate change. In addition to existing methods that use toxic solvents, electrochemical techniques that can work with sustainable electricity are now becoming available. The TU Delft research group led by David Vermaas worked together with Wetsus and Caltech to analyze these sustainable technologies for CO2 removal, and compared them for the first time. The researchers also described which methods show the most potential for making large-scale CO2 removal possible. Their paper on this was published recently in the scientific journal Energy & Environmental Science.
It sounds like an ideal solution to combat climate change, but CO2 removal is really not so easy. There are various circumstances under which CO2 can be captured. It can take place directly at a large source of CO2, but it is also important to be able to capture CO2 from decentralized emission point sources. Researcher David Vermaas explains: "Ultimately, power plants and industry are only responsible for a certain proportion of carbon emissions. Decentralized sources, such as cars, agricultural industries, heating and aircraft together account for 40% of CO2 emissions in the atmosphere."
Electrochemical routes
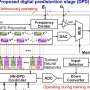
Methods have been developed for both centralized and decentralized capture, but for decentralized capture in particular, there are few energy-efficient, clean methods available. A possible solution is provided by what are known as electrochemical routes, which enable the pH of a solution to be adapted and the CO2 to be concentrated. Laboratory tests have proved that these techniques are effective and work on the basis of electricity rather than heat. Here the CO2 is converted into carbonate or bicarbonate salts, and then concentrated as pure CO2 or limestone.
Vermaas's research group has now compared the various electrochemical methods for the first time, and studied their feasibility. Their studies showed that for all the methods, the energy required forms a barrier. Vermaas: "In theory not much energy is required to convert CO2 into another form of carbonate or bicarbonate, but right now the current methods are not efficient enough. On average 50% more energy is used than is needed in a theoretical conversion." The relatively low concentration of CO2 in the atmosphere means it has proved particularly difficult to use it as feedstock for the conversion reaction.
Ocean as a buffer
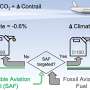
Vermaas and his colleagues are using their analysis to steer the direction of future research. One option that shows great potential is capturing CO2 from the ocean. Vermaas states, "The ocean acts as a huge buffer absorbing large amounts of CO2 from the atmosphere every year, and that makes this the ultimate way to capture CO2 from decentralized sources." We could adapt existing desalination plants, so that in the future they can also remove CO2 from seawater. This concept based on acids and bases has become more feasible and energy-efficient in recent times through the use of membranes with improved conductivity and improved electrochemical cell designs. An additional benefit is that this technique prevents lime scale deposits in the desalination plants.
Vermaas's research group is already working on developing the technology in which the CO2 in the water is converted into either pure CO2 gas or calcium carbonate. Working on a scale of 10 cm, they have already succeeded in carrying out the necessary reactions in the lab. Now they are looking step-by-step at how to apply the technology on a larger scale. Vermaas: "We are seeing—and rightly so—that the urgency of the climate crisis means there is more attention from both governmental bodies and the business community for the development and scaling up of carbon capture methods. This is a good thing, because ultimately it is essential that we bring the capture and conversion of CO2 closer."
More information: R. Sharifian et al. Electrochemical carbon dioxide capture to close the carbon cycle, Energy & Environmental Science (2020). DOI: 10.1039/D0EE03382K