Improved water splitting method: A green energy innovation
Having used fossil fuels for over a century for nearly everything, humanity has triggered a climate crisis. Now, the directive is to achieve net zero emissions or carbon neutrality by 2050.
A hydrogen economy is one way in which a carbon neutral world can thrive. At present, the simplest way to produce hydrogen fuel is electrochemical water splitting: Running electricity through water in the presence of catalysts (reaction-enhancing substances) to yield hydrogen and oxygen. This reaction, however, is very slow, requires specialized conditions and noble-metal catalysts, and is, overall, expensive. Thus, achieving a high hydrogen yield in an energy-efficient manner at low cost is challenging. To date, hydrogen production from water splitting has not been successfully commercialized.
Now, a team of researchers from Pusan National University, Korea, led by Professor Kandasamy Prabakar, have developed a method to design a novel electrocatalyst that can solve some of these problems. Their work was made available online on April 6, 2021, and will be published in print in the September 2021 issue of Volume 292 of Applied Catalysis B: Environmental.
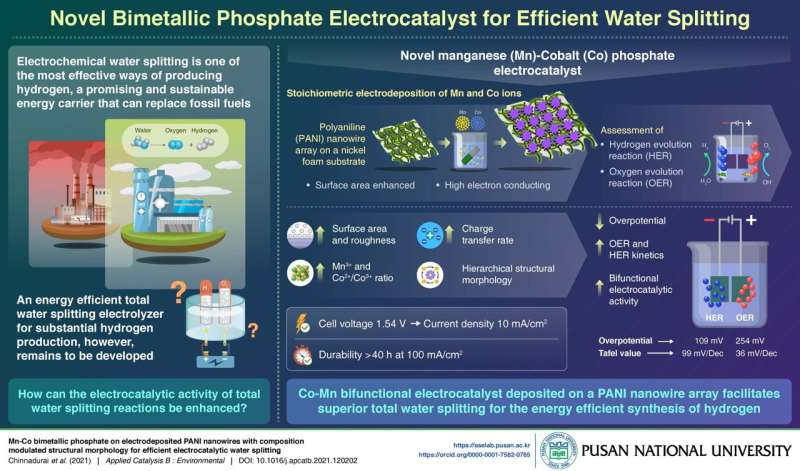
Describing the study, Prof. Prabakar says, "Today, 90% of hydrogen is produced from steam reforming processes that emit greenhouse gases into the atmosphere. In our laboratory, we have developed a non-noble metal based stable electrocatalyst on a polymer support which can effectively produce hydrogen and oxygen from water at a low-cost from transition metal phosphates."
Prof. Prabakar's team fabricated this electrolyzer by depositing cobalt and manganese ions, in varying proportions, on a Polyaniline (PANI) nanowire array using a simple hydrothermal process. By tuning the Co/Mn ratio, they have achieved an overall high surface area for the reactions to occur, and combined with the high electron conducting capacity of the PANI nanowire, faster charge and mass transfer was facilitated on this catalyst surface. The bimetallic phosphate also confers bifunctional electrocatalytic activity for the simultaneous production of oxygen and hydrogen.
In experiments to test the performance of this catalyst, they found that its morphology substantially decreases the reaction overpotential, thereby improving the voltage efficiency of the system. As a testament to durability, even after 40 hours of continuous hydrogen production at 100 mA/cm2, its performance remains consistent. And water splitting was possible at a low input voltage of merely 1.54V.
In addition to these advantages, is the low cost of transition metals. Indeed, the system can be scaled and adapted for application to myriad settings. Speaking of possible future applications, Prof. Prabakar explains, "Water-splitting devices that use this technology can be installed onsite where hydrogen fuel is required, and can function using a low energy input or a completely renewable source of energy. For instance, we can produce hydrogen at home for cooking and heating using a solar panel. This way, we can achieve carbon neutrality well before 2050."
More information: Deviprasath Chinnadurai et al, Mn-Co bimetallic phosphate on electrodeposited PANI nanowires with composition modulated structural morphology for efficient electrocatalytic water splitting, Applied Catalysis B: Environmental (2021). DOI: 10.1016/j.apcatb.2021.120202