December 1, 2021 feature
A molecular platform for heterogeneous OER electrocatalysis based on double-atom catalysts
The oxygen evolution reaction (OER) is a chemical reaction that enables the generation of oxygen gas. This reaction is crucial for producing fuels through water splitting or through the electroreduction of CO2.
So far, mixed metal oxides containing Co, Fe or Ni have been found to be the most promising electrocatalysts for eliciting this reaction. However, due to their heterogeneity, these catalysts and their reaction mechanisms can be difficult to study.
In recent years, scientists have also been closely examining atomically dispersed catalysts, an emerging class of heterogeneous electrocatalysts with a high atomic efficiency, which include single-atom catalysts and discrete sub-nano clusters. This new type of catalyst is characterized by uniform and well-defined active sites, which might be easier to probe and examine.
Researchers at Ecole Polytechnique Federale de Lausanne (EPFL) and National Taiwan University have recently introduced a new molecular platform that could be used to prompt and study heterogeneous OER electrocatalysis. This platform, introduced in a paper published in Nature Energy, is based on double-atom catalysts, catalysts in which all active metal species exist as two defined atoms.
"Although a few atomically dispersed OER catalysts were recently reported, the structures of their active sites during OER remained unclear," Hao Ming Chen, one of the researchers who carried out the study, told TechXplore. "In our previous work, we showed that we could transform a single-atom Co pre-catalyst into a Co-Fe double atom catalyst for OER in-situ. This catalyst exhibits one of the highest turnover frequencies among metal oxides."
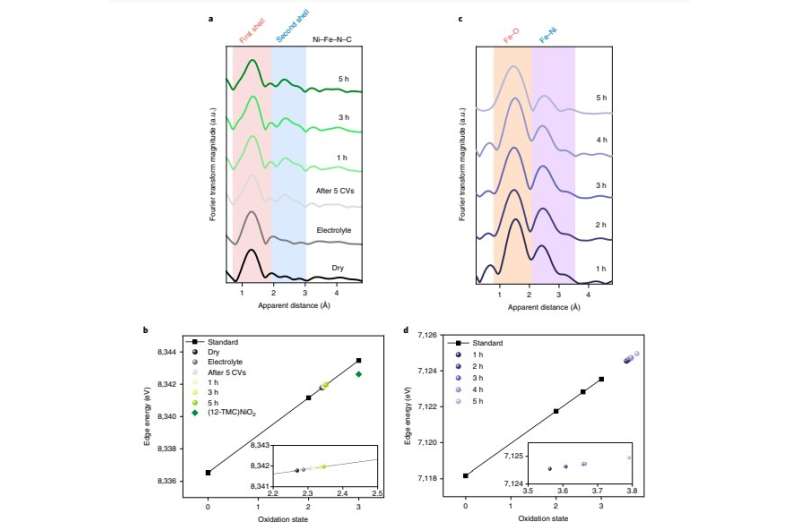
Using a technique known as operando X-ray absorption spectroscopy (XAS), Chen and his colleagues showed that the active site of the catalyst they created was a dimeric Co-Fe moiety. Their findings ultimately proved the potential of double-atom molecular platforms for the development of defined and highly active OER catalysts.
"We believe that one of the most important subjects in heterogeneous oxygen evolution electrocatalysis today is to develop new approaches that facilitate the understanding of active heterogeneous catalysts," Chen said. "However, in our previous work, we only demonstrated the Co-Fe double-atom catalysts."
To prove the overall effectiveness of double-atom catalysts for heterogeneous OER electrocatalysis, the researchers showed that the transformation into double-atom catalysts is possible for Co, Fe, and Ni containing single-atom pre-catalysts. They thus created various double-atom catalysts and characterized their structures using operando XAS and high-energy-resolution fluorescence detected (HERFD) XAS. In addition, they examined them using a method known as aberration-corrected scanning transmission electron microscopy analysis.
"These iso-structural and defined catalysts provide a rare molecular framework for the mechanistic study of heterogeneous OER catalysts," Chen said. "The double-atom catalysts presented here can be considered as molecular models of the active site of mixed metal oxides, which are among the most active catalysts for OER."
The recent work by Chen and his colleagues introduces double-atom catalysts as a new valuable platform for OER catalysis. In addition, the researchers outlined a simple and straightforward method for synthesizing these catalysts.
"By utilizing operando X-ray absorption studies and performing electrokinetic analyses on all catalysts, we can deduce catalytic cycles for OER, which is extremely challenging and has not been realized to date," Chen said. "Because our catalysts have molecularly defined active site, they bridge molecular and heterogeneous solid-state catalysts. The corresponding mechanistic study should have a far-reaching impact."
In the future, the new molecular platform proposed by this team of researchers could be used to study heterogenous OER electrocatalysts more in depth. For instance, it could help material scientists to better understand how a catalyst's atomic configuration and chemical state affect its catalytic nature.
"We believe that our work will inspire numerous follow-up studies employing double-atom catalysis as a new design strategy and may also be applicable to other heterogeneous electrocatalysts," Chen added. "For example, in the electrochemical CO2 reduction, it can be speculated that the C-C coupling has to occur on the catalytic surface to form the C-C bonding formation for generating C2 products, such as C2H4 and C2H5OH. We are planning to develop the double-atom electrocatalysts for heterogeneous electrochemical CO2 reduction."
More information: Lichen Bai et al, Double-atom catalysts as a molecular platform for heterogeneous oxygen evolution electrocatalysis, Nature Energy (2021). DOI: 10.1038/s41560-021-00925-3
© 2021 Science X Network