Sodium-based material yields stable alternative to lithium-ion batteries

University of Texas at Austin researchers have created a new sodium-based battery material that is highly stable, capable of recharging as quickly as a traditional lithium-ion battery and able to pave the way toward delivering more energy than current battery technologies.
For about a decade, scientists and engineers have been developing sodium batteries, which replace both lithium and cobalt used in current lithium-ion batteries with cheaper, more environmentally friendly sodium. Unfortunately, in earlier sodium batteries, a component called the anode would tend to grow needle-like filaments called dendrites that can cause the battery to electrically short and even catch fire or explode.
In one of two recent sodium battery advances from UT Austin, the new material solves the dendrite problem and recharges as quickly as a lithium-ion battery. The team published their results in the journal Advanced Materials.
"We're essentially solving two problems at once," said David Mitlin, a professor in the Cockrell School of Engineering's Walker Department of Mechanical Engineering and Applied Research Laboratory who designed the new material. "Typically, the faster you charge, the more of these dendrites you grow. So if you suppress dendrite growth, you can charge and discharge faster, because all of a sudden it's safe."
Graeme Henkelman, a professor in the Department of Chemistry and the Oden Institute for Computational Engineering and Sciences, used a computer model to explain, from a theoretical perspective, why the material has the unique properties it does.
"This material is also exciting because the sodium metal anode theoretically has the highest energy density of any sodium anode," Henkelman said.
Demand is rising for stationary energy storage systems for homes and for smoothing out the ebb and flow of wind and solar energy on electric grids. At the same time, lithium mining has been criticized for its environmental impacts, including heavy groundwater use, soil and water pollution and carbon emissions. Lithium-ion batteries typically also use cobalt, which is expensive and mined mostly in the Democratic Republic of Congo, where it has significant impacts on human health and the environment. By comparison, sodium mining is cheaper and more environmentally friendly.
Mitlin is bullish on the idea that this new innovation and others from UT Austin, including a new solid electrolyte that boosts energy storage, will mean sodium batteries may soon be able to fill the growing demand for stationary energy storage.
When a rechargeable battery is being charged, ions (such as lithium or sodium) move from one component called the cathode to another called the anode. When the battery is being used to generate electricity, the ions move from the anode back to the cathode.
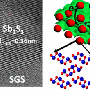
The new anode material, called sodium antimony telluride intermetallic – Na metal composite (NST-Na), is made by rolling a thin sheet of sodium metal onto an antimony telluride powder, folding it over on itself, and repeating many times.
"Think of making a kind of layered pastry, like spanakopita," Mitlin said.
This process results in a very uniform distribution of sodium atoms that makes it less likely to form dendrites or surface corrosion than existing sodium metal anodes. That makes the battery more stable and allows faster charging, comparable to a lithium-ion battery's charge rate. It also has a higher energy capacity than existing sodium-ion batteries.
Henkelman said that if the sodium atoms that carry a charge in a sodium battery bind more strongly to each other than they do to the anode, they tend to form instabilities, or clumps of sodium that attract more sodium atoms and eventually lead to dendrites. He used a computer simulation to reveal what happens when individual sodium atoms interact with the new composite material NST-Na.
"In our calculations, this composite binds sodium a little more strongly than sodium binds itself, which is the ideal case for having the sodium atoms come down and evenly spread out on the surface and prevent these instabilities from forming," Henkelman said.
The study's two lead authors Yixian Wang and Hui Dong—current and former graduate students in Mitlin's lab respectively—fabricated the material. Colleagues at Los Alamos National Laboratory led by John Watt characterized its properties. The study's other authors are Hongchang Hao, Pengcheng Liu and Naman Katyal of UT Austin.
Mitlin, Wang and Dong have applied for a patent, along with UT Austin, on the new sodium metal anode material's fabrication, structure and functionality.
More information: Yixian Wang et al, A Sodium–Antimony–Telluride Intermetallic Allows Sodium‐Metal Cycling at 100% Depth of Discharge and as an Anode‐Free Metal Battery, Advanced Materials (2021). DOI: 10.1002/adma.202106005