Hydrogen detection using fiber optic sensors
Hydrogen plays a pivotal role in Germany's energy and climate policy. In comparison to other gaseous or liquid energy sources, special security requirements have to be considered for applications with hydrogen, because additionally to fire hazard under certain conditions an explosive air hydrogen mixture can arise from leaks in tanks or pipelines. To further increase safety levels when dealing with hydrogen, researchers at the Fraunhofer Institute for Telecommunications, Heinrich-Hertz Institute, HHI are working on fiber-optic-based sensors that can detect hydrogen and are superior to conventional sensors in many respects.
To achieve the climate targets that have been set and to prevent increased global warming, all countries have to reduce the consumption of fossil fuels in their energy mix budgets as quickly as possible. There is a growing focus on hydrogen technologies as a sustainable alternative—especially in the industrial production and mobility sectors. Wherever hydrogen is used, stored, transported and transferred, appropriate safety precautions must be taken into account. Even though hydrogen is not toxic, weighs less than air and thus rises to the top, dangerous situations can occur: Indeed, if the hydrogen concentration in the air exceeds a threshold of four percent, which can happen quickly if there is sufficient pressure in a hydrogen tank or if a room is not properly ventilated, a small ignition source, a single spark, is enough to trigger an explosion.
Small, easy to integrate and with no inherent safety risk
An anticipatory approach is needed to prevent this, and Dr. Günter Flachenecker, Senior Scientist at Fraunhofer HHI, knows how to go about this. At the Fiber Optical Sensor Systems branch lab department of the Fraunhofer HHI in Goslar, the physicist and his team are for new technological approaches to detect hydrogen using glass fiber sensors: "Conventional, commercially available safety sensors for sensing hydrogen, which are typically catalytic heat tone sensors or electrochemical cells, require an electrical power supply. In the worst-case scenario, both variants could act as a source of ignition and trigger the explosion that they are supposed to prevent, if the device or the electrical supply lines have a defect," explains Flachenecker. "Our fiber optic sensors principally do not have this risk. At the same time, they don't require complex wiring, are small and can be easily integrated into a wide variety of structures in the plant or vehicle to be monitored."
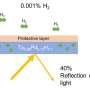
Optical fibers are virtually predestined for sensory applications in a safety-related environment because they are robust and have only a small diameter of around a quarter of a millimeter. For an optical fiber to become a hydrogen sensor, it needs to be modified in a number of places. To do this, a laser is firstly used to imprint certain structures into the fiber optic core, creating what is known as a fiber Bragg grating—a periodic refractive index modulation that ensures that light is reflected at a certain wavelength.
A special functional coating is then applied around the glass fiber sensory part to ensure that the glass fiber reacts specifically to hydrogen: "We work with catalytic layers, for example palladium or palladium alloys," Flachenecker says. "Palladium has the ability to soak up hydrogen, much like a sponge. As soon as the two substances meet, the hydrogen disintegrates into its atomic fragments and the hydrogen atoms that are released penetrate the crystal framework of the palladium. This causes elongation in the optical fiber, which can be measured instantaneously as a change in the reflected light signals via the built-in fiber Bragg grating. As soon as the hydrogen concentration in the air drops again, the hydrogen is released from the palladium." This means that there is no damage to the coating and the sensor can be reused. Flachenecker goes on to emphasize that the process described here only works because hydrogen atoms are very small. Other substances cannot penetrate the palladium layer in this way.
Potential for a wide range of applications
This is not, however, the only method that the researchers have tested. For instance, it is also possible to detect hydrogen with glass fibers whose fiber claddings have been etched away, or with a very thin layer of nanoparticles applied to the glass fiber surface. "It's a big playing field, and there are a lot of things we want to try out," Flachenecker says. "It's critical that we find ways of detecting hydrogen that are fast enough to prevent accidents and that respond reliably within the required sensitivity range. In that regard, we are certainly making good progress at the moment."
In practice, for example, the new fiber optic sensors could become an integral part of hydrogen-powered vehicles and be used to monitor hydrogen refueling stations, auto repair shops or electrolyzers. Based on this technology a larger sensor network can be scaled up easily that monitors hydrogen infrastructure at many points simultaneously. The electronics for recording the measurement data, for example a spectrometer for optical evaluation of the fiber optic sensors, could be installed in a safe place at any distance from the sensors. If a certain concentration of hydrogen is exceeded and the sensor picks this up, then the alarm management system for the relevant application is triggered and specific measures, such as an acoustic warning signal, the closing of valves or the opening of windows, can be initiated within a few seconds.
The ongoing research project led by Günter Flachenecker is funded by the German Federal Ministry for Economic Affairs and Climate Action and is being carried out in cooperation with a local fire protection company. It started two years ago and will conclude in the summer when an ongoing field test, in which the fiber optic sensors are being fitted in trucks, will come to an end. There are plans for a follow-up project that will see the new sensors tested in even greater detail and further preparatory steps taken towards certification and commercialization. The goal is clear: To able to work with hydrogen in an even safer way without causing any accidents.