Scientists apply control theory in analysis of photochemical reaction dynamics in light therapies
Researchers in China have recently applied control theory for the first time in analyzing the dynamics of the photochemical reactions in light therapies, and developed the first mathematical model of the light-induced singlet oxygen in treating fungal infections. The results were published in IEEE Transactions on Biomedical Engineering.
Photodynamic therapies (PDT) are non-antibiotic alternatives for treating localized infectious diseases due to their rapid action and lack of drug resistance. Similar to PDT, blue light therapies that rely only on the endogenous pigments, i.e., porphyrins and flavins, of the pathogens are also effective and even safer to use.
Anti-fungal blue light (ABL) has been widely studied as a new treatment approach of fungal infections. Calculating the quantum yield of singlet oxygen is the key to determine the light dosage applied in both PDT and ABL.
In order to investigate the dynamics of the photosensitized oxidation reactions in PDT and ABL, and to develop an effective modeling approach, Dr. Dong Jianfei's team at the Suzhou Institute of Biomedical Engineering and Technology (SIBET) of the Chinese Academy of Sciences has applied fundamental control theory to analyze these processes, and derived the linearization condition for the nonlinear first-principle PDT model.
The mechanism of the anti-fungal effect of PDT and ABL is light exciting either the exogenous photosensitizer in the former case or the endogenous pigments in the latter, which in turn produces reactive oxygen species (ROS) from triplet oxygen (3O2) molecules.
ROS are highly reactive, and can cause cytotoxicity. Singlet oxygen (1O2) usually accounts for 80% of all the ROS induced by light; while hydroxyl radicals and other types of ROS take the remaining 20%. Furthermore, singlet oxygen is a precursor of most other ROS.
Dong and his colleagues linearized the nonlinear first-principle PDT model at a set of equilibrium points along the trajectory of its dynamic response to light stimuli.
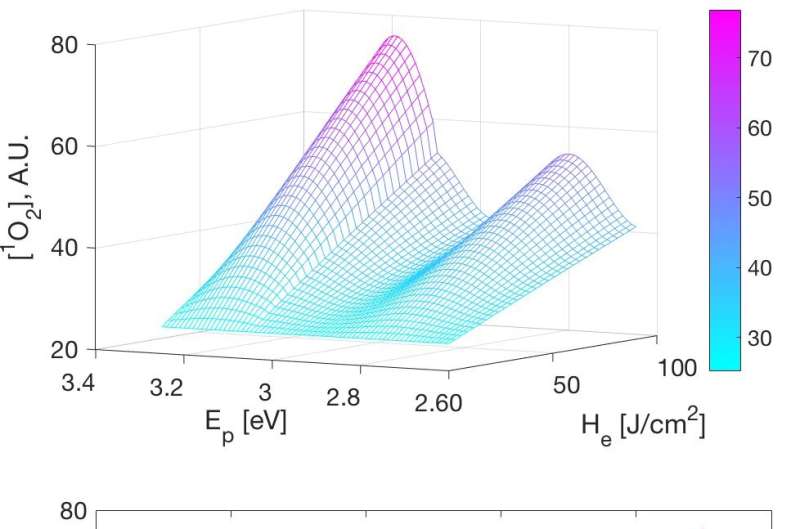
This resulted in a set of linear-time-invariant (LTI) state-space models. These models are all of third order, and contain three poles and one zero. Among these, one pole is constantly located at the origin of the complex plane.
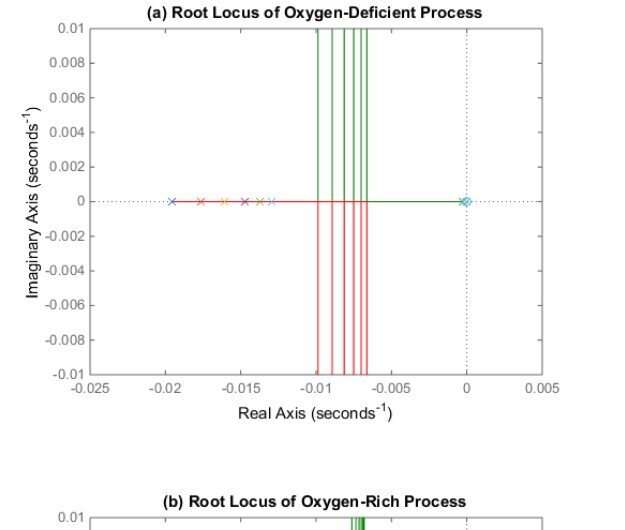
Further analysis showed that the zero can approximately cancel the pole at the origin, leading to second-order models containing two poles. The locations of these two remaining poles are highly related to the concentration of 3O2, i.e., the main ingredient to produce 1O2.
According to the researchers, when the concentration of 3O2 is ample, the root loci at the different points are condensed to a much smaller cluster, than those in the oxygen-deficient case. This indicates that the local LTI systems in the oxygen-rich case are more identical to each other; and that is to say, the original nonlinear first-principle PDT model tends to be linear.
"This is an interesting observation," said Dong. In fact, the oxygen concentration in the blood is sufficient for photochemical reactions in most cases.
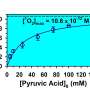
Inspired by this, the team further analyzed and solved the first-principle PDT model, and obtained a closed-form analytical solution under the oxygen-rich condition. The significance of this analytical solution is that it is a nonlinear algebraic equation with only four parameters, which can be easily fitted to experimental data.
They further proposed a data-driven modeling approach for the photochemical reaction process of light therapies. The model has achieved a good fitting result on the measured ABL experimental data.
It is the first attempt of applying control theory to analyze the photochemical reaction dynamics of light therapies in terms of their nonlinearity. The proposed modeling techniques also offer opportunities for determining the light dosages in treating fungal infection diseases, especially those on the surface tissues of human body.
More information: Tianfeng Wang et al, Analysis and data-based modeling of the photochemical reaction dynamics of the induced singlet oxygen in light therapies, IEEE Transactions on Biomedical Engineering (2022). DOI: 10.1109/TBME.2022.3170541