This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
The novel chemistry behind ultra-high power density batteries

Mohammad Asadi, assistant professor of chemical engineering at Illinois Institute of Technology, has published a paper in the journal Science describing the chemistry behind his novel lithium-air battery design. The insights will allow him to further optimize the battery design, with the potential for reaching ultra-high power densities far beyond current lithium-ion technology.
The battery design has the potential to store one kilowatt-hour per kilogram or higher—four times greater than lithium-ion battery technology, which would be transformative for electrifying transportation, especially heavy-duty vehicles such as airplanes, trains, and submarines.
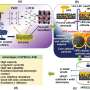
Asadi aimed to make a battery with a solid electrolyte, which provides safety and energy benefits compared to liquid electrolyte batteries, and sought an option that would be compatible with the cathode and anode technologies that he has been developing for use in lithium-air batteries.
He chose a mix of polymer and ceramic, which are the two most common solid electrolytes but both of which have downsides. By combining them, Asadi found that he could take advantage of ceramic's high ionic conductivity and the high stability and high interfacial connection of the polymer.
The result allows for the critical reversible reaction that enables the battery to function—lithium-dioxide formation and decomposition—to occur at high rates at room temperature, the first demonstration of this in a lithium-air battery.
As described in the Science paper, Asadi has conducted a range of experiments that demonstrate the science behind how this reaction occurs.
"We found that that solid-state electrolyte contributes around 75 percent of the total energy density. That tells us there is a lot of room for improvement because we believe we can minimize that thickness without compromising performance, and that would allow us to achieve a very, very high energy density," says Asadi.
These experiments were conducted in collaboration with University of Illinois Chicago and Argonne National Laboratory. Asadi says he plans to work with industry partners as he now moves toward optimizing the battery design and engineering it for manufacturing.
"The technology is a breakthrough, and it has opened up a big window of possibility for taking these technologies to the market," says Asadi.
More information: Alireza Kondori et al, A room temperature rechargeable Li2O-based lithium-air battery enabled by a solid electrolyte, Science (2023). DOI: 10.1126/science.abq1347