This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Research reveals thermal instability of solar cells but offers a bright path forward
A new type of solar technology has seemed promising in recent years. Halide perovskite solar cells are both high performing and low cost for producing electrical energy—two necessary ingredients for any successful solar technology of the future. But new solar cell materials should also match the stability of silicon-based solar cells, which boast more than 25 years of reliability.
In newly published research, a team led by Juan-Pablo Correa-Baena, assistant professor in the School of Materials Sciences and Engineering at Georgia Tech, shows that halide perovskite solar cells are less stable than previously thought. Their work reveals the thermal instability that happens within the cells' interface layers, but also offers a path forward towards reliability and efficiency for halide perovskite solar technology.
Their research, published as the cover story for the journal Advanced Materials in December 2022, has immediate implications for both academics and industry professionals working with perovskites in photovoltaics, a field concerned with electric currents generated by sunlight.
Lead halide perovskite solar cells promise superior conversion of sunlight into electrical power. Currently, the most common strategy for coaxing high conversion efficiency out of these cells is to treat their surfaces with large positively charged ions known as cations.
These cations are too big to fit into the perovskite atomic-scale lattice, and, upon landing on the perovskite crystal, change the material's structure at the interface where they are deposited. The resulting atomic-scale defects limit the efficacy of current extraction from the solar cell. Despite awareness of these structural changes, research on whether the cations are stable after deposition is limited, leaving a gap in understanding of a process that could impact the long-term viability of halide perovskite solar cells.
"Our concern was that during long periods of solar cell operation the reconstruction of the interfaces would continue," said Correa-Baena. "So, we sought to understand and demonstrate how this process happens over time."
To carry out the experiment, the team created a sample solar device using typical perovskite films. The device features eight independent solar cells, which enables the researchers to experiment and generate data based on each cell's performance. They investigated how the cells would perform, both with and without the cation surface treatment, and studied the cation-modified interfaces of each cell before and after prolonged thermal stress using synchrotron-based X-ray characterization techniques.
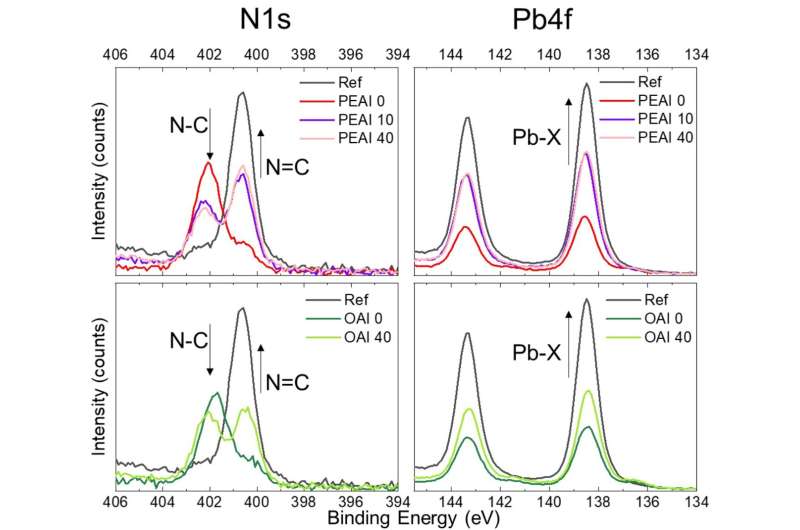
First, the researchers exposed the pre-treated samples to 100 degrees Celsius for 40 minutes, and then measured their changes in chemical composition using X-ray photoelectron spectroscopy. They also used another type of X-ray technology to investigate precisely what type of crystal structures form on the film's surface. Combining the information from the two tools, the researchers could visualize how the cations diffuse into the lattice and how the interface structure changes when exposed to heat.
Next, to understand how the cation-induced structural changes impact solar cell performance, the researchers employed excitation correlation spectroscopy in collaboration with Carlos Silva, professor of physics and chemistry at Georgia Tech. The technique exposes the solar cell samples to very fast pulses of light and detects the intensity of light emitted from the film after each pulse to understand how energy from light is lost. The measurements allow the researchers to understand what kinds of surface defects are detrimental to performance.
Finally, the team correlated the changes in structure and optoelectronic properties with the differences in the solar cells' efficiencies. They also studied the changes induced by high temperatures in two of the most used cations and observed the differences in dynamics at their interfaces.
"Our work revealed that there is concerning instability introduced by treatment with certain cations," said Carlo Perini, a research scientist in Correa-Baena's lab and the first author of the paper. "But the good news is that, with proper engineering of the interface layer, we will see enhanced stability of this technology in the future."
The researchers learned that the surfaces of metal halide perovskite films treated with organic cations keep evolving in structure and composition under thermal stress. They saw that the resulting atomic-scale changes at the interface can cause a meaningful loss in power conversion efficiency in solar cells. In addition, they found that the speed of these changes depends on the type of cations used, suggesting that stable interfaces might be within reach with adequate engineering of the molecules.
"We hope this work will compel researchers to test these interfaces at high temperatures and seek solutions to the problem of instability," Correa-Baena said. "This work should point scientists in the right direction, to an area where they can focus in order to build more efficient and stable solar technologies."
More information: Carlo Andrea Riccardo Perini et al, Interface Reconstruction from Ruddlesden–Popper Structures Impacts Stability in Lead Halide Perovskite Solar Cells, Advanced Materials (2022). DOI: 10.1002/adma.202204726