This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Expert discusses how polymer solid-state batteries could be used in e-aircraft
Solid-state batteries are considered as beacon of hope in battery research because, among other things, they can store higher amounts of energy and are faster to charge than conventional liquid electrolytes.
The head of the department for Methodology and Electrolyte Research PD Dr. Gunther Brunklaus from Helmholtz Institute Münster (HI MS; IEK-12) of Forschungszentrum Jülich explains in an interview why he is placing a special focus on polymer electrolyte materials, what still needs to be done before they are deemed ready for the market and what applications are in prospect for solid-state batteries.
What hopes are currently being discussed with regard to solid-state batteries, what are their potentials?
Gunther Brunklaus: One hope comprises above all that higher amounts of energy can be stored in solid electrolytes with comparable amounts of material than in conventional battery systems with liquid electrolytes. This means that longer ranges can be achieved in mobile applications, for example. Less material may result in lower CO2 emissions in production and faster charging than with liquid electrolytes is also possible.
Why could higher amounts of energy be stored?
Brunklaus: For example, other anode materials, such as lithium metal, which enables considerably higher specific capacity compared to typically applied graphite anodes, can be exploited. On the cathode side, there is the possibility to utilize materials that have not previously worked with liquid electrolytes. Incidentally, solid-state batteries are already being used in applications such as pacemakers and smart watches.
With these applications, the topic of safety immediately comes to mind. Are there also advantages here?
Brunklaus: Yes. For one thing, highly volatile liquid materials can be dispensed with. This reduces the risk of possible spontaneous combustion as well as the general fire hazard, since liquid components can escape more quickly in an accident, for example, causing short circuits in the cells unlike solid components.
In mechanical tests with nail impact on battery cells, it was even shown that individual polymer solids in particular are rather safe due to self-healing properties, due to which they might isolate the source of the fire through chemical reactions, thus stopping the fire while allowing the battery cell to continue operation. Also, safety aspects can be taken into account when selecting electrode materials that are not suitable for liquid electrolytes.
For what other applications are solid-state batteries conceivable?
Brunklaus: First of all, I would like to say that there is no such thing as the one ideal cell chemistry for all possible application scenarios. Each application offers its own challenges, so different solutions have their justification. In addition to their current use in the medical field and in smart watches, solids are suitable for vehicle batteries in the context of electromobility, for utilization in stationary energy storage, for example as buffer capacity in renewable energy generation, as well as for cell phones and power tools.
Polymer-based battery cells are already exploited today, for example in electrified busses. Vehicle batteries should be easy and fast to charge, whereas stationary storage units may have a slower charging speed. Other properties are important here, for example, low-cost and environmentally friendly production. In case of cell phones, a compromise between fast charging and high charging capacity in a small space is required.
What is the current focus of research in your department?
Brunklaus: We are developing methods to better understand charge transfer processes or other chemical processes that counteract battery longevity or operation. These include electrochemical impedance spectroscopy and magnetic resonance methods. All of this together helps us to develop various cell designs, each of which opens up its own application options. At the Münster site of HI MS, we also focus strongly on the production and optimization of polymer electrolytes and protective coatings.
What can polymer electrolytes do in contrast to other solids?
Brunklaus: They offer the advantage that they are flexible in shape and could be used, for example, as so-called structural battery in aviation. It would be conceivable to install them in the wall of an e-aircraft. Due to their chemical variability, polymers have a practically unlimited reservoir of different structures and functional groups and thus offer numerous options for adaptation to the required properties of possible applications.
Solid ceramic electrolytes, on the other hand, could break in an aircraft. The flowable properties of polymers serve, so to speak, to fill any gaps in the battery cell system that porous electrodes made of carbon or sulfur-carbon composite material, for example, might have. Unused volumes and high charge transfer resistances are hence avoided.
What are the biggest challenges you face in the development of polymer electrolytes?
Brunklaus: There are two major construction sites. First, the ionic conductivity—important for high performance and fast charging—must be further increased. The macroscopic conductivity of the main electrolyte, however, is not the only decisive factor here, but specifically the charge transfer dynamics at the interfaces to the electrode, which must enable high currents during rapid charging of the cells.
Second, the material must be scaled up cost-efficiently into the kilogram range for certain applications, such as stationary storage, without sacrificing quality, and options for processing of the materials during cell production also must be adapted. In a foreseeable future, flowable components such as shorter-chain oligomers will likely be needed in the cathode area to afford high current densities. To work on these many construction sites, cooperation across different locations is required.
Which cooperations exist?
Brunklaus: Within the framework of the BMBF competence cluster FestBatt, for example, Helmholtz Institute Münster, Karlsruhe Institute of Technology (KIT), Helmholtz Institute Ulm (HIU), as well as the University of Duisburg-Essen and other renowned institutions are cooperating to develop scientific and technical solutions for solid-state batteries.
Overall, the aim is to open up the polymer world, so to speak, as well as the areas of other material classes, and to realize as many variants of battery cell systems as possible in terms of application options. To this end, we are bringing together our materials expertise in the fields of polymers, hybrids, ceramics, cell designs and analytics. The coupling of theory and experiment is also essential and is supported by our collaborations.
For example, the practical macroscopic methods are well complemented by computer simulations in the microscopic range, also including 'machine learning' approaches.
More information: Yi‐Hsuan Chen et al, Green Polymer Electrolytes Based on Polycaprolactones for Solid‐State High‐Voltage Lithium Metal Batteries, Macromolecular Rapid Communications (2022). DOI: 10.1002/marc.202200335
Louise Frenck et al, Failure Mechanisms at the Interfaces between Lithium Metal Electrodes and a Single-Ion Conducting Polymer Gel Electrolyte, ACS Applied Materials & Interfaces (2022). DOI: 10.1021/acsami.2c16869
Gerrit Michael Overhoff et al, Ceramic-in-Polymer Hybrid Electrolytes with Enhanced Electrochemical Performance, ACS Applied Materials & Interfaces (2022). DOI: 10.1021/acsami.2c13408
Min-Huei Chiou et al, Durable fast-charging lithium metal batteries designed with cross-linked polymer electrolytes and niobate-coated cathode, Journal of Power Sources (2022). DOI: 10.1016/j.jpowsour.2022.231528
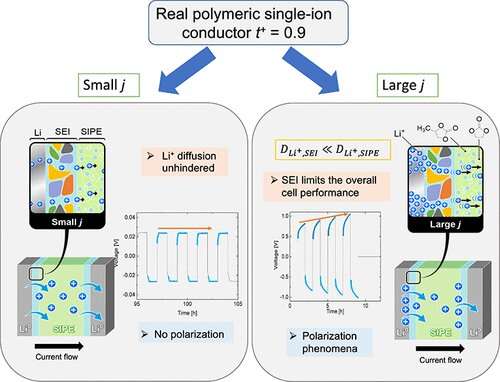
Kristina Borzutzki et al, Does Cell Polarization Matter in Single-Ion Conducting Electrolytes?, ACS Applied Materials & Interfaces (2022). DOI: 10.1021/acsami.1c19097