May 8, 2023 dialog
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
written by researcher(s)
proofread
Solar cells inspire Li-ion batteries
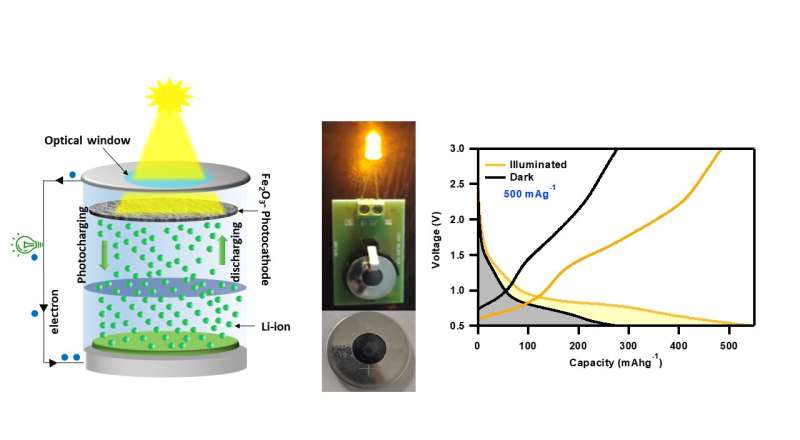
Solar energy is at the forefront of the global shift toward producing sustainable energy sources and addressing energy poverty. However, the intermittent nature of solar energy limits its use for applications such as IoT devices, live remote sensing and an off-grid power supply. Conventionally the batteries are powered by solar cells to store the energy for later use. However, the physical combination of these two technologies requires separate packaging of both systems, is cumbersome to install, and requires more electrodes, which increases the cost and ohmic losses in the device.
Moreover, these physically connected photovoltaic (PV) panels and batteries make use of different types of energy materials to achieve both energy harvesting and storage, which makes the overall system bulky. As a result, these issues limit applications.
In this context, the demonstrated photorechargeable batteries (PRBs) can provide a promising solution to overcome the limitations associated with the physical integration of PVs and batteries. A PRB can perform solar energy harvesting and storage simultaneously in a single device using advanced nanomaterials, which can perform energy harvesting and storage efficiently. This cutting-edge technology promises to be lightweight and efficient compared to the existing conventional combination of PVs and batteries.
Light-induced conversion mechanism for PRBs
In their study published in Advanced Sustainable Systems, researchers at the Advanced Energy Materials Lab, Department of Physics at the Indian Institute of Technology Jodhpur have demonstrated that iron oxide (also known as hematite) nanorods can work as an active material to form efficient and low-cost photocathodes for PRB applications. The high theoretical specific capacity (1006 mAh g-1), earth abundance, nontoxicity, environmental friendliness and low processing techniques make the alpha phase of iron oxide an attractive anode material for lithium-ion batteries.
The iron oxide nanorods have shown the capability to simultaneously harvest the solar radiation in the visible region due to their bandgap of ~2.1 eV and store the Li-ions efficiently. This work provides the first demonstration of standalone photocharging by exploring the conversion reaction mechanism where more than 90% enhancement in the specific capacity of the lithium-ion battery is achieved upon solar illumination.
"The highly nanoporous photocathodes are fabricated using hematite, C-61 carbon (PCBM) and carbon nanotubes. Hematite can absorb sunlight and produce photogenerated charge carriers, while PCBM and carbon nanotubes conductive additives provided a suitable pathway for photogenerated electrons to reach the current collector and initiate photocharging," said Shubham Chamola, the first author of the research article.
The nanoporous iron oxide nanorods are fabricated by using a low-cost solution processing technique in which the carbon nanotubes are functionalized with PCBM and blended with the NMP solution of hematite nanoparticles, which resulted in the growth of Fe2O3 nanorods on carbon nanotubes bundles. To assemble the photorechargeable battery, the researchers used a modified 2032 coin cell casing with an 8 mm diameter hole punched on the bottom to serve as an optical window to couple light. This window was sealed with a transparent PVC sheet and the active material based photocathode was placed facing toward the window.

The PRB showed independent charging when illuminated with a 470 nm blue LED, achieving a photoconversion and storage efficiency (PCSE) of 1.988%, which is a significant achievement in the field of PRBs compared to the earlier published results based on the intercalation-based ion storage. Under white light LED illumination at a high current rate of 2,000 mA g-1, the PRB showed a 92.96% enhancement in specific capacities. When the PRB is exposed to light, Fe2O3 nanorods absorb photons of energy higher than their energy band gap and generate photogenerated charge carriers at the photocathode. The conductive additives provide a favorable pathway for photoelectrons to reach the current collector and further the anode through an external circuit.
Simultaneously, photoholes present in Fe2O3 oxidize Fe0 to Fe3+ which provides repulsion to the Li+ toward the Li-metal anode via the electrolyte. As a result, the Li-ions are reduced at the anode to form Li-metal, resulting in photocharging. This phenomenon was further demonstrated by recording the OCV under a resistive discharge and when the light was turned ON, the OCV began to increase—a phenomenon that is contrary to what would normally be expected. This suggests that more photogenerated charge carriers are present than are required to meet the current demand for discharging the PRB.
This phenomenon could be beneficial where continuous operation of sensors is required, making PRBs capable of charging during daylight hours and using the stored energy to power the devices at night or when there is no sunlight. The underlying mechanism was further studied by performing various electrochemical measurements, such as OCV analysis under different resistances, electrochemical impedance spectroscopy, and cyclic voltammetry under dark and illuminated conditions. Furthermore, the PRB was able to power a commercial 3V LED even after three months of fabrication, showing that the demonstrated iron oxide nanorods-based PRB are highly stable and do not suffer from self-discharge issues.
"PRBs hold significant promises for the futuristic energy solution; however, this technology is still in its infancy and requires extensive research for the development of efficient materials and an understanding of the mechanism at an atomistic level before it can compete with the established integrated Si solar cells and Li-ion battery technology," said Dr. Shahab Ahmad, the principal investigator of the project.
This story is part of Science X Dialog, where researchers can report findings from their published research articles. Visit this page for information about ScienceX Dialog and how to participate.
More information: Shubham Chamola et al, High Performance Photorechargeable Li‐Ion Batteries Based on Nanoporous Fe2O3 Photocathodes, Advanced Sustainable Systems (2023). DOI: 10.1002/adsu.202300043
Dr. Shahab Ahmad is currently working as a faculty member at the Department of Physics, IIT Jodhpur and leading the "Advanced Energy Materials Group." Prior to this, he has worked extensively on energy materials and devices at the University of Cambridge, U.K. His research areas include Li-ion batteries, photo-rechargeable Li-ion batteries, metal halide perovskites for photovoltaic applications and solar assisted water splitting process for H2 production.
Mr. Shubham Chamola is a PhD student working with Dr. Shahab Ahmad at IIT Jodhpur, his current area of research includes the development of high performance photo rechargeable energy storage devices.
More information about their research activities can be found here: sites.google.com/view/shahabahmad1/home?authuser=0















