This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New method could break down PFAS left on water treatment filters
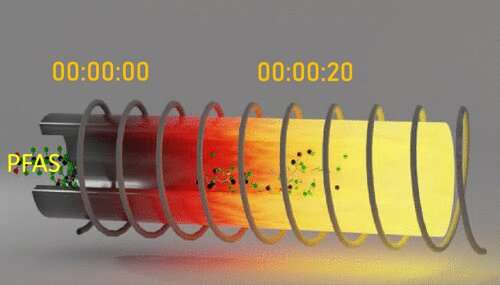
In a recent study published in ACS ES&T Engineering, Feng "Frank" Xiao and colleagues at the University of Missouri demonstrate an innovative method using thermal induction heating to rapidly break down PFAS left on the surface of two solid materials—granular activated carbon and anion exchange resins—after these materials have been used to filter PFAS from municipal water systems. The team's goal is to clean the materials before they are properly disposed.
PFAS is a group of synthetic chemicals commonly found in household and industrial products such as firefighting foam, food packaging and nonstick cookware. The method is based on the Joule heating effect, which uses the process of electromagnetic induction inside a metallic reactor.
"In this study, we explored the use of an engineering technique used to melt metals," Xiao said. "This method produced 98% degradation of PFAS on the surface of absorbents like granular activated carbon and anion exchange resins after just 20 seconds, which makes this process highly energy efficient and much faster than conventional methods."
In recent years, experts have raised concerns about the risks to human health from environmental exposure to PFAS, including development of cancer and other serious health issues. Xiao, whose appointment is in the Department of Civil and Environmental Engineering, said while PFAS can be filtered out of water using adsorbents, the disposal of used or "spent" adsorbents also creates issues of environmental contamination.
"Since the group of chemicals known as PFAS generally resist degradation, they pose considerable challenges to established treatment processes, including the waste disposal practices for materials used as filters like granular activated carbon and anion exchange resins," Xiao said.
Xiao has spent his career focused on researching ways to safely remove PFAS from the environment, including recently demonstrating similar efficiency with the use of induction heating to rapidly degrade PFAS in soil. He said the current study also drew inspiration from recent proposed regulation by the Environmental Protection Agency (EPA) that, if finalized, would require public water systems in the U.S. to monitor and reduce PFAS contamination in drinking water and spent adsorbents.
Potential drawbacks of this method include by-products created during this process—organic fluorinated species and hydrogen fluoride. While these by-products are considered toxic to consume through breathing or ingestion, Xiao has a solution.
"If the gaseous organic fluorinated products are not degraded during induction heating, abatement treatment will be necessary to remove or degrade them," Xiao said. "However, based on my previous studies, some of these products can be degradable by regular thermal approaches. Simultaneously, the generation of hydrogen fluoride is increased, which is desirable because it means greater mineralization, or decomposition, of PFAS. We've found hydrogen fluoride can be removed simply using clay or soil at moderate temperatures."
More information: Feng Xiao et al, Thermal Phase Transition and Rapid Degradation of Forever Chemicals (PFAS) in Spent Media Using Induction Heating, ACS ES&T Engineering (2023). DOI: 10.1021/acsestengg.3c00114