This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Design strategies for rechargeable aqueous metal-ion batteries
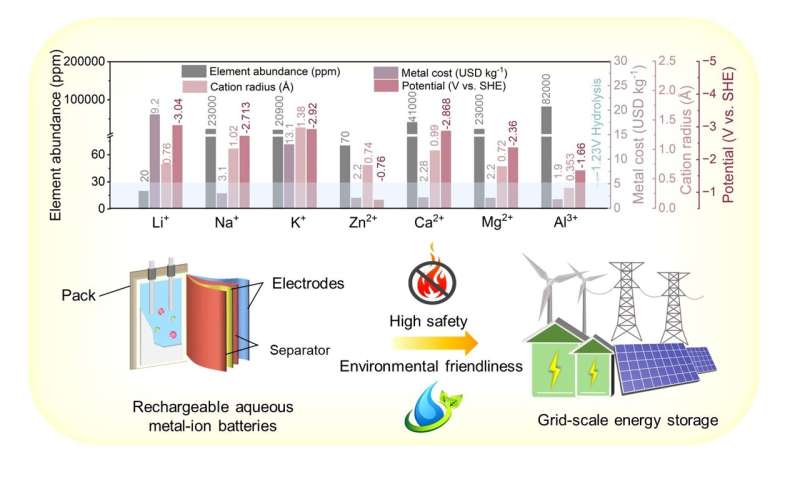
Rechargeable aqueous metal-ion batteries (AMBs) have attracted extensive scientific and commercial interest due to their potential for cost-effective, highly safe, and scalable stationary energy storage. However, their limited output voltage, inadequate energy density, and poor reversibility of ambiguous electrode reactions in aqueous electrolytes strongly limits their practical viability.
This review, led by Prof. Feiyu Kang and Prof. Dong Zhou (Tsinghua Shenzhen International Graduate School), aims to elucidate the challenges of existing AMBs from material design to whole de-vice applications. They summarize the emerging electrochemistry, fundamental properties, and key issues in interfacial behavior of various classes of prevailing AMBs, including aqueous alkali metal-ion batteries and multivalent-ion batteries, and present an appraisal of recent advances for addressing the performance deficiency.
Specifically, the progress of zinc-ion batteries is highlighted to provide a ubiquitous guideline for their commercialization in grid-scale energy storage.
Although thousands of articles have been reported on this hot area, great efforts are still needed to overcome obstacles and promote the commercial application of AMBs:
(1) Electrode Materials. It is imperative for the rational design and/or modification of high-energy, electrochemically stable and cost-effective electrode materials toward practical applications. For the cathode materials, the ambiguous carrier storage mechanism remains to be clarified via spatially resolved and/or nondestructive in-situ characterization techniques. On the anode side, efficient suppression of interfacial side reactions (e.g., corrosion, hydrogen evolution reaction and/or dendrite formation) by surface modification and anode structure/composition design is of key significance to realize high CE, satisfied active material utilization and long lifespan.
(2) Aqueous electrolytes. The narrow electrochemical window of aqueous electrolytes is the main cause of the low energy density of AMBs. The design principles for wide-temperature-range electrolytes should be conducted based on their inherent advantages of non-flammability, high ionic conductivity, and cost-effectiveness.
(3) Road to commercialization. Ultrathin separators, corrosion-resistant current collectors, low-expansion conductive additives and stable binders are highly important for the long-term operation of practical AMBs. However, these key battery components have long been overlooked in laboratory research.
Although the overall cost of AMBs may be estimated to be less than many current rechargeable battery chemistries, future cost analyses must consider both the anode (e.g., Zn foil) thickness and the cathode's area capacity to realize practical optimization schemes that can be used for scaling up production. In addition, the ecological toxicity of the materials used in the batteries must also be considered, especially in the context of handling or recycling waste batteries.
The findings are published in the journal Science China Chemistry.
More information: Yang Li et al, Design strategies for rechargeable aqueous metal-ion batteries, Science China Chemistry (2023). DOI: 10.1007/s11426-023-1698-6