This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Research team develops highly efficient solar cell that enables decentralized production of hydrogen
Researchers around the world are working on more efficient methods for producing hydrogen. Hydrogen could make a decisive contribution to reducing the consumption of fossil raw materials, especially if it is produced using renewable energies. Existing technologies for producing climate-neutral hydrogen are still too inefficient or too expensive for broader application.
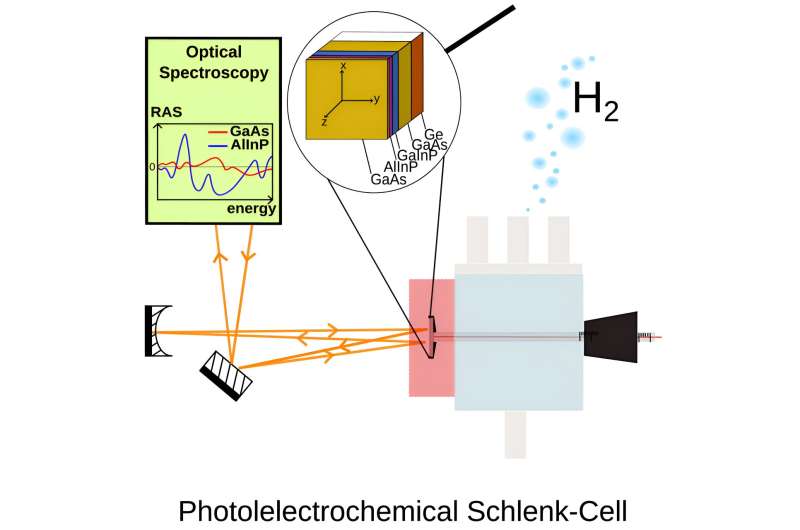
A research team at the University of Tübingen is now presenting a new type of solar cell with remarkably high efficiency. It enables decentralized production of hydrogen and has the potential for industrial-scale applications. The results have been published in the journal Cell Reports Physical Science.
A solar cell on a dive
When hydrogen is produced from water via electrolysis using renewable energy, it is referred to as "green hydrogen" because of its climate-friendly production. In solar water splitting, often referred to as artificial photosynthesis, hydrogen is produced using energy from the sun.
A research team led by Dr. Matthias May from the Institute of Physical and Theoretical Chemistry at the University of Tübingen has developed a solar cell that is an integral part of the photoelectrochemical apparatus and works directly with the catalysts for water splitting. The special feature of the Tübingen development is that an additional external circuit, such as in a photovoltaic solar panel, is no longer necessary.
This innovative approach makes the technology more compact, more flexible and potentially more cost-efficient. But with this design come greater demands on the solar cell. "Among researchers in the field, realizing stable and efficient photoelectrochemical or direct water splitting is something of a holy grail," May says.
The special feature of the solar cell structure is the high degree of control of the interfaces between the different materials. The surface structures here are produced and checked on a scale of a few nanometers, or millionths of a millimeter. Particularly troublesome are small crystalline defects that occur, for example, during the growth of solar cell layers. Such defects also change the electronic structure and may therefore reduce both the efficiency and the stability of the system.
May adds, "Overall, however, corrosion—and thus the long-term stability of the solar cell in water—is still the greatest challenge. In this, we have now made great progress compared to our earlier work."
The technical design of the new cell is both innovative and particularly effective. The efficiency rate of the cell indicates what percentage of the energy from sunlight can be converted into usable hydrogen energy (calorific value).
The research team has achieved an efficiency of 18%, the second highest value ever measured for direct solar water splitting. The first significant efficiencies for solar water splitting were presented in 1998 by NREL in the U.S. with 12%. This was followed only in 2015 by a jump to 14% and in 2018 to 19%.
Several ventures at other universities demonstrate that the technology, even with much lower efficiencies, can be commercialized. Erica Schmitt, first author of the study, explains, "What we have developed here is a technology for solar hydrogen production that does not require a high-performance connection to the electricity grid. This means that permanent smaller stand-alone solutions for energy supply are also conceivable."
The next steps include improving long-term stability, transferring to a more cost-effective silicon-based material system and scaling up to larger surface areas. The research results could make a significant contribution to energy supply and the reduction of CO2 emissions.
More information: Erica A. Schmitt et al, Photoelectrochemical Schlenk cell functionalization of multi-junction water-splitting photoelectrodes, Cell Reports Physical Science (2023). DOI: 10.1016/j.xcrp.2023.101606


















