This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Unlocking the mystery behind the performance decline in a promising cathode material—implications for EV batteries
The first generation of lithium-ion batteries for electric vehicles has been a remarkable success story. Yet, the question arises: What changes to battery materials will spur further advances to extend driving range and lower costs?
A better positive electrode, or cathode, for lithium-ion batteries has been the focus of intense past research. The cathode is one of the main components in batteries. Several candidates for cathode materials offer the prospect of batteries with much higher energy storage, leading to longer driving range. However, the capacity, or amount of current flowing out within a given time, tends to decline rapidly with charge-discharge cycling for reasons unknown.
Researchers at the U.S. Department of Energy's (DOE) Argonne National Laboratory have discovered the main reason why and how one of the more promising cathode materials degrades with use. That material is a lithium nickel manganese cobalt (NMC) oxide rich in nickel and in the form of single nanosized crystals. In single crystals, all the atoms are arranged in the same highly ordered pattern.
"Nickel-rich NMC is especially appealing because it uses 70-80% nickel, a high-capacity material, and requires much less cobalt," said Liu. Cobalt is expensive and considered a critical mineral because of supply issues.
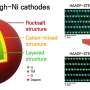
Typically, the nickel-rich NMC cathode consists of particles of multiple crystalline forms, or polycrystals, randomly oriented with respect to each other. With charge-discharge cycling, however, these clusters crack at the boundaries among the crystals, and the cathode capacity rapidly drops.
It had been hypothesized that fabricating the cathode with single crystals instead of polycrystals would solve the cracking problem, as the boundaries would be eliminated. However, even single-crystal cathodes failed prematurely, leaving scientists perplexed.
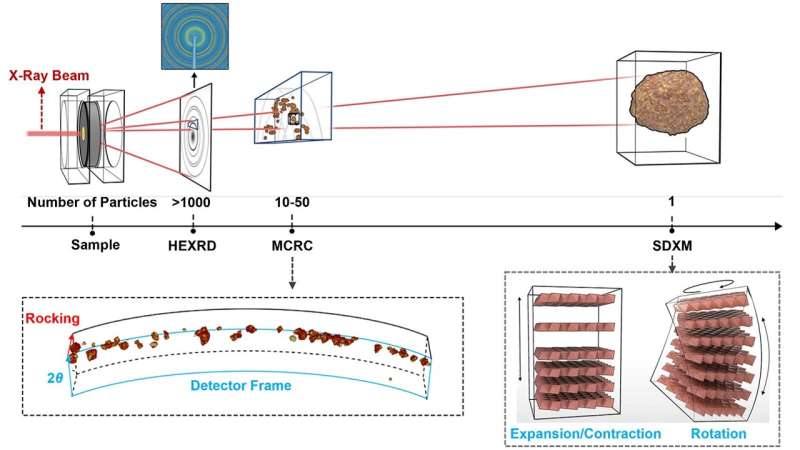
To uncover the mechanism, the team devised a pioneering method that combines multiscale X-ray diffraction and high-resolution electron microscopy. These materials analyses were done at the Advanced Photon Source (APS) at Argonne, the National Synchrotron Light Source at DOE's Brookhaven National Laboratory and Argonne's Center for Nanoscale Materials (CNM). All three are DOE Office of Science user facilities.
"The problem with electron microscopy alone is that it only provides a snapshot of a small area on a single crystal," said Materials Scientist Tao Zhou in CNM. "And while X-ray diffraction offers insights into internal structures of many particles, it lacks surface-level information. Our method bridges this gap, offering a comprehensive understanding at the scale of one, 10 to 50, and 1,000 particles."
The atoms in single crystals are arranged in neatly ordered rows and columns called lattices. The team's multifaceted analyses of single-crystal cathodes provided crucial information about changes in the lattice on charge and discharge.
As Liu and Zhou explained, introduction of a charge triggers a strain on the lattice that causes it to expand and rotate, disrupting the neatly ordered pattern of atoms. Upon discharge, the lattice contracts to its original state, but the rotation remains. With repeated charge-discharge cycles, the rotation becomes more pronounced. This change in the cathode structure causes a steep performance drop.
Critical to gaining these insights were measurements with the Hard X-ray Nanoprobe operated jointly by CNM and APS.
"The team's new method was instrumental in understanding the burning issue of why nickel-rich NMC cathodes with single crystals fail so rapidly," said Khalil Amine, an Argonne Distinguished Fellow. "This newfound understanding will give us ammunition to fix this issue and enable lower-cost electric vehicles with longer driving range."
"Our method should also be useful to understanding failure mechanisms in other battery types than present-day lithium-ion," added Liu.
This research appeared in Science. In addition to Liu, Zhou and Amine, authors include Weiyuan Huang, Lei Yu, Jing Wang, Junxiang Liu, Tianyi Li, Rachid Amine, Xianghui Xiao, Mingyuan Ge, Lu Ma, Steven N. Ehrlich, Martin V. Holt and Jianguo Wen.
More information: Weiyuan Huang et al, Unrecoverable lattice rotation governs structural degradation of single-crystalline cathodes, Science (2024). DOI: 10.1126/science.ado1675