Proof-of-concept nanogenerator turns CO₂ into sustainable power
University of Queensland researchers have built a generator that absorbs carbon dioxide (CO2) to make electricity.
Apr 18, 2024
2
149
Electronics & Semiconductors

University of Queensland researchers have built a generator that absorbs carbon dioxide (CO2) to make electricity.
Apr 18, 2024
2
149
Energy & Green Tech

For more than two millennia, paper has been a staple of human civilization. But these days, the use of paper is not limited to writing. It is also playing a pivotal role in ushering in a greener future.
Apr 3, 2024
0
35
Electronics & Semiconductors
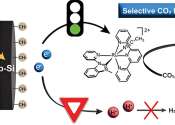
Researchers in the UNC-Chapel Hill Chemistry Department are using semiconductors to harvest and convert the sun's energy into high-energy compounds that have the potential to produce environmentally friendly fuels.
Apr 2, 2024
0
74
Business

Lower-income communities across the United States have long been much slower to adopt solar power than their affluent neighbors, even when local and federal agencies offer tax breaks and other financial incentives.
Mar 28, 2024
0
34
Engineering
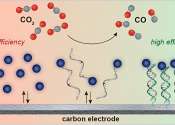
MIT chemical engineers have devised an efficient way to convert carbon dioxide to carbon monoxide, a chemical precursor that can be used to generate useful compounds such as ethanol and other fuels.
Mar 27, 2024
0
175
Energy & Green Tech

Researchers have successfully transformed CO2 into methanol by shining sunlight on single atoms of copper deposited on a light-activated material, a discovery that paves the way for creating new green fuels.
Mar 25, 2024
0
210
Electronics & Semiconductors

Researchers have built a new kind of two-faced (bifacial) panel. They used single-walled carbon nanotubes as both front and back electrodes.
Mar 18, 2024
0
122
Energy & Green Tech
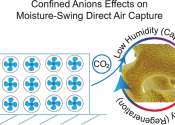
In a corner of Kelsey Hatzell's lab sits a small jar filled with a material that has an ability far beyond what its nondescript appearance would suggest: a way to capture and release carbon dioxide from the atmosphere by ...
Mar 15, 2024
1
82
Energy & Green Tech
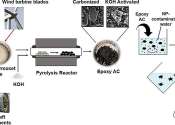
University of Waterloo researchers have created a new technology that can remove harmful nanoplastics from contaminated water with 94% efficiency. The study, "Utilization of epoxy thermoset waste to produce activated carbon ...
Mar 14, 2024
0
526
Electronics & Semiconductors

Small wearable or implantable electronics could help monitor our health, diagnose diseases, and provide opportunities for improved, autonomous treatments. But to do this without aggravating or damaging the cells around them, ...
Mar 13, 2024
0
19
Carbon (pronounced /ˈkɑrbən/) is the chemical element with symbol C and atomic number 6. As a member of group 14 on the periodic table, it is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. There are three naturally occurring isotopes, with 12C and 13C being stable, while 14C is radioactive, decaying with a half-life of about 5730 years. Carbon is one of the few elements known since antiquity. The name "carbon" comes from Latin language carbo, coal, and, in some Romance and Slavic languages, the word carbon can refer both to the element and to coal.
There are several allotropes of carbon of which the best known are graphite, diamond, and amorphous carbon. The physical properties of carbon vary widely with the allotropic form. For example, diamond is highly transparent, while graphite is opaque and black. Diamond is among the hardest materials known, while graphite is soft enough to form a streak on paper (hence its name, from the Greek word "to write"). Diamond has a very low electrical conductivity, while graphite is a very good conductor. Under normal conditions, diamond has the highest thermal conductivity of all known materials. All the allotropic forms are solids under normal conditions but graphite is the most thermodynamically stable.
All forms of carbon are highly stable, requiring high temperature to react even with oxygen. The most common oxidation state of carbon in inorganic compounds is +4, while +2 is found in carbon monoxide and other transition metal carbonyl complexes. The largest sources of inorganic carbon are limestones, dolomites and carbon dioxide, but significant quantities occur in organic deposits of coal, peat, oil and methane clathrates. Carbon forms more compounds than any other element, with almost ten million pure organic compounds described to date, which in turn are a tiny fraction of such compounds that are theoretically possible under standard conditions.
Carbon is one of the least abundant elements in the Earth's crust, but the fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. It is present in all known lifeforms, and in the human body carbon is the second most abundant element by mass (about 18.5%) after oxygen. This abundance, together with the unique diversity of organic compounds and their unusual polymer-forming ability at the temperatures commonly encountered on Earth, make this element the chemical basis of all known life.
This text uses material from Wikipedia, licensed under CC BY-SA