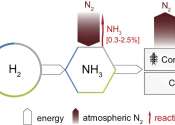
Nitrogen (pronounced /ˈnaɪtrədʒɨn/) is a chemical element that has the symbol N and atomic number 7 and atomic mass 14.00674 u. Elemental nitrogen is a colorless, odorless, tasteless and mostly inert diatomic gas at standard conditions, constituting 78% by volume of Earth's atmosphere.
Many industrially important compounds, such as ammonia, nitric acid, organic nitrates (propellants and explosives), and cyanides, contain nitrogen. The extremely strong bond in elemental nitrogen dominates nitrogen chemistry, causing difficulty for both organisms and industry in converting the N2 into useful compounds, and releasing large amounts of energy when these compounds burn or decay back into nitrogen gas.
The element nitrogen was discovered by Daniel Rutherford, a Scottish physician, in 1772. Nitrogen occurs in all living organisms. It is a constituent element of amino acids and thus of proteins, and of nucleic acids (DNA and RNA). It resides in the chemical structure of almost all neurotransmitters, and is a defining component of alkaloids, biological molecules produced by many organisms.