Increasing the efficiency of fuel cells could lead to more widespread use of zero-emissions vehicles

On an average weekday, the air in Beijing can be so choked with smog that entire buildings disappear from view. Emissions from heavy industry and coal power plants are largely to blame, but so too are the nearly 5 million cars and trucks that clog the city's streets each morning, throwing a fog of toxic particulates into the atmosphere. Vehicle emissions like these are a growing public health problem in China and other developing nations, and it's unlikely to improve any time soon.
Widespread use of electric vehicles could offer some relief from the smog, says Iryna Zenyuk, but driving an electric car any significant distance requires charging dozens of heavy batteries, a process that can take hours. "That's very uncomfortable for an end user who's used to filling up at the pump and rolling away five minutes later," says Zenyuk, an associate professor of mechanical engineering at Tufts.
The answer, Zenyuk says, is to develop hydrogen fuel cells—a cleaner, more efficient power source that converts hydrogen gas into electricity through a series of electrochemical reactions. Building fuel cells that are both efficient and affordable, however, has proved extremely difficult. Zenyuk and her colleagues are working to understand and overcome a key stumbling block to creating a commercially viable product.
Fuel Cell CAT Scans
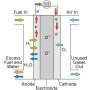
The reactions fuel cells use to generate power are relatively simple and robust. As hydrogen enters a fuel cell, it passes through a porous layer made of a polymer and platinum-covered carbon. This layer strips electrons off each atom, leaving only protons behind. The protons then pass through a membrane and end up at another similar layer called a cathode, where they join together with oxygen atoms, forming molecules of water. In the process, the cell creates an electric charge that can power a car.
The difficulty is with the water resulting from that last step. While it's a clean byproduct, which makes the technology attractive from an environmental perspective, it eventually compromises efficiency. As droplets form inside a fuel cell, Zenyuk says, they slowly plug up the pores of its cathode layer, stopping oxygen in the air from entering. The less oxygen that makes it inside the cell, the less power the cell generates. Even small amounts of water, when stuck inside a cathode's pores, can block enough oxygen to have a big impact on the efficiency of the device.
"Essentially, once the water is formed, you have this liquid filling the cathode layer, and you have to remove it fast," she says. "Water management is one of the critical problems with fuel cells. You need sufficient water to hydrate the membrane, but too much of it will flood the cell."
At the moment, many fuel cell manufacturers build channels in the cathode that help wick water away from open pores. It's not a perfect system, however, because no one knows how water is distributed in the tiniest pores of the cathode layer. Until researchers can figure that out, they'll be hard-pressed to build a fuel cell that sheds the liquid effectively.
Zenyuk and her team are chipping away at the problem by developing new ways to look inside the cathodes. If they can see how minuscule droplets of water initially form within them, she reasons, they might be able to tailor-make a structure that spits out the liquid before it collects into large droplets.

Getting the images they need while a cell is running isn't a simple task, however. In order to see water forming inside the tiny pores of a fuel cell's cathode, Zenyuk's team must enlist the help of a synchrotron—basically a gigantic particle accelerator—to bombard the cell with intense beams of x-rays.
"We essentially run a miniaturized fuel cell in the cyclotron, then rotate it slowly by 180 degrees. For each degree, we collect an image. It's almost like giving it a CAT scan," she says.
By combining images taken at different angles, her team builds a detailed 3-D model of water droplets as they're created in holes just a few microns across—far smaller than the width of a human hair.
The Long Game
Although these images are starting to shed light on how water forms inside a fuel cell, Zenyuk warns that researchers are still a long way off from being able to use that information to improve a cell's efficiency. At the moment, her team is still working on improving the resolution of their images, and honing their techniques to see if they'll work on full-sized fuel cells.
Even when they do get reliable data, she says, creating a new cathode material that can reliably shed water droplets won't be easy. The cathode is responsible for a number of key jobs inside a fuel cell—it must be strong enough to offer structural support, conductive enough to carry an electric charge, and porous enough for gases to flow through it. Add water management to the list, and you've got one serious engineering challenge.
"These are complex materials," Zenyuk says, "There's not just one problem you have to tackle when you're designing them. There are usually two or three problems in a combined approach. It's all intertwined. When you work with fuel cells, you're dealing with chemistry coupled to materials science coupled to engineering. It takes a really interdisciplinary approach."
Zenyuk has a long history of tackling complex challenges. After graduating college, she spent a year playing competitive chess, ultimately becoming one of the top 10 female chess masters in the country. She says the mindset needed to play the game at a high level—studying a new problem, picking it apart and thinking strategically—has come in handy in the lab.
So while she knows it could be a while before you'll be able to pick up a can of hydrogen at the corner store, or pull your car up to a hydrogen pump for a top-off, that doesn't seem to bother her. As in her chess-playing days, Zenyuk's got her eye on the long game. Thanks in part to her work, fuel cells could transform from niche market items into ubiquitous sources of energy over the next decade—not just for cars, but for consumer electronics as well. In the near future, she notes, small fuel cells might be used to power computers, appliances or cell phones.