June 28, 2018 report
Researchers use micro-robots to carry cells to a target site in live animals

A team of researchers at the City University of Hong Kong have found a way to use micro-robots to carry a cluster of cells to a target site in live animals. In their paper published in the journal Science Robotics, the group describes the micro-robots they designed and their performance during testing.
In recent years, scientists have begun looking into the feasibility of using micro-robots to carry drugs or other cargo from one part of the body to another. Such micro-robots could deliver medication, to a specific part of the body that needs it, for example, rather than to the whole body, preventing unwanted side-effects. In this new effort, the researchers were looking for a way to carry a cluster of cells from one location to another inside of a living animal. If such an approach proved possible, micro-robots could be used to carry stem cells to injured body parts, for example, to repair them.
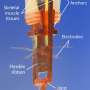
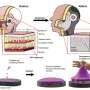
To design a micro-robot suitable for the task, the researchers started by running simulations on a computer looking for the optimal shape. They found that one shaped like a burr (as in, a spiky seed pod) worked the best of all they tried. They used a 3-D printer to create micro-robots with room for a pouch inside for holding stem cells and connective tissue. Then they covered the micro-robots with nickel to make them magnetic, and with titanium for biocompatibility. To move the micro-robot from one place to another, they used a magnet.
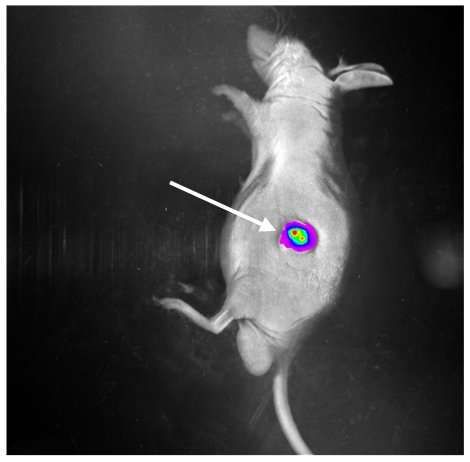
They tested their micro-robot idea by injecting several into zebrafish embryos and then successfully manipulating them externally with a tiny magnet. They then filled one of their micro-robots with fluorescent cancer cells and injected it into a live mouse and guided it to a new site where the cancer cells were released. After monitoring the mouse for a few weeks, they found that the micro-robot had successfully released its load—the new location glowed, indicating that the cancer cells had taken hold. The researchers plan to continue their research and expect to begin clinical trials sometime over the next five years.

More information: Junyang Li et al. Development of a magnetic microrobot for carrying and delivering targeted cells, Science Robotics (2018). DOI: 10.1126/scirobotics.aat8829
Abstract
The precise delivery of targeted cells through magnetic field–driven microrobots/carriers is a promising technique for targeted therapy and tissue regeneration. This paper presents a microrobot designed with a burr-like porous spherical structure for carrying and delivering targeted cells in vivo under a magnetic gradient field–driven mechanism. The robot was fabricated by using three-dimensional laser lithography and coated with Ni for magnetic actuation and Ti for biocompatibility. Numerical and experimental studies demonstrated that the proposed microrobot design could enhance magnetic driving capability, promote cell-carrying capacity, and benefit cell viability. Microrobots loaded with cells could be automatically controlled to reach a desired site by using a self-constructed electromagnetic coil system, as verified by in vivo transport of cell-cultured microrobots in zebrafish embryos. The carried cells could be spontaneously released from the microrobot to the surrounding tissues; in vitro experiments showed that cells from the microrobot were directly released onto the desired site or were able to pass through the blood vessel–like microchannel to arrive at the delivery area. Further in vivo cell-releasing tests were performed on nude mice, followed by histological study. This research provides a microrobotic device platform for regenerative medicine and cell-based therapy.
© 2018 Tech Xplore