January 20, 2020 feature
Gradient Li-rich oxide cathode particles for batteries with minimum oxygen release
Lithium-rich transition metal oxide (Li1+XM1-XO2) cathodes have potential for use in Li-ion batteries, powering electronic devices and electric vehicles. These cathodes have a high energy density, typically above 900 Wh kg-1, yet they currently also come with significant limitations.
The most crucial problem observed in most Li-rich cathodes is that they release oxygen to electrolytes, and thus, their voltage decays while they are being used. This significant limitation has prevented their widespread use for years.
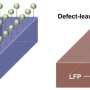
Researchers at the Massachusetts Institute of Technology (MIT) have recently devised a strategy that could help to overcome this issue, immunizing Li-rich oxide cathode particles against oxygen release. This new strategy, outlined in a paper published in Nature Energy, involves a molten salt treatment that eliminates the release of oxygen from Li-rich single crystals to electrolytes by making the surface region Li-poor, while still enabling stable oxygen redox contributions inside the particles.
"Our main objective was to utilize oxygen's capacity for redox reactions without making the reduced oxygen ions (i.e., "peroxo" and "superoxo"-like) globally mobile, which means that they can escape from the cathode particle surface and react with the electrolyte inside a battery," Ju Li, one of the researchers who carried out the study, told TechXplore.
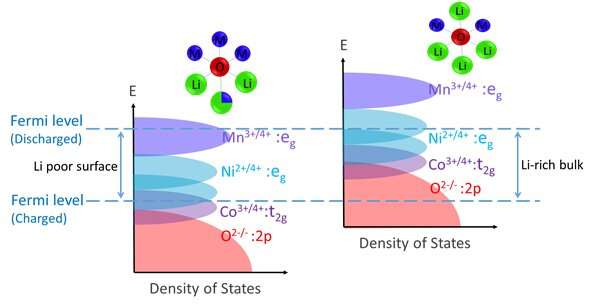
The reduced oxygen ions inside Li-rich cathodes somewhat resemble metal-peroxo and metal-superoxo complexes; compounds through which blood transports oxygen in animals. The peroxo (O-) and superoxo (O0.5-) species, while contributing capacity, have much higher mobility than the standard O2-. In Li1+XM1-XO2 cathodes. These oxygen ions can move around freely and ultimately escape from the cathode particles, reacting with and contaminating the liquid electrolyte.
To prevent this from happening, Li and his colleagues implemented a treatment involving the extraction of lithium oxide (LiO) using molten molybdate salt at high temperatures. They found that this treatment allows the surface to gain the composition Li1-X'M1+X'O2 without disrupting the lattice continuity or creating excess defects (epitaxial), thus removing peroxo (O-) and superoxo (O0.5-) species near the surface, preventing the Li-rich single crystals from releasing oxygen to electrolytes.
"We performed an immunization treatment, so a surface regions about ~10nm thick are depleted of oxygen, and therefore would be extra stable in battery cycling," Li said. "The immunization treatment was performed at a high temperature of 700°C, so as we extract oxygen and lithium the lattice repairs itself by thermal annealing and goes smoothly from Li-rich to Li-poor, without additional defects and without losing the perfect lattice coherence of the single-crystalline particle."
The immunization strategy devised by Li and his colleagues does not affect the metal valence states and the structure of Li-rich crystals inside the cathode, thus maintaining a stable oxygen anion-redox (O2-↔O-) capacity contribution while a battery is operating. In tests evaluating their strategy, the researchers found that it resulted in a gradient hybrid anion- and cation-redox (HACR) cathode with a specific density or 843 Wh kg-1 after 200 cycles at 0.2C and 808 Wh kg-1 after 100 cycles at 1C, with a minimum oxygen release and thus lower consumption of the electrolyte in the battery.
"Our study proves that cycling a battery full cell with very little amount of electrolyte (industrial level of 2g(electrolyte)/Ah) is possible, indicating that we have stopped oxygen loss while utilizing the oxygen's redox capacity," Li said. "This so-called 'solid oxygen' battery concept has the potential to double the energy density of cathodes."
By reducing the oxygen release typically observed in Li-rich cathodes, the strategy devised by Li and his colleagues could eventually facilitate the commercialization and widespread use of lithium-based batteries powered by these cathodes. Interestingly, the immunization treatment outlined in their study could also be applied to other elements, helping to suppress or prevent unexpected surface reactions in batteries. In their next studies, the researchers plan to scale up the synthesis in the Li-rich cathode-based battery and further improve the compressed density of HACR cathodes.
More information: Zhi Zhu et al. Gradient Li-rich oxide cathode particles immunized against oxygen release by a molten salt treatment, Nature Energy (2019). DOI: 10.1038/s41560-019-0508-x
© 2020 Science X Network