Thermal stability analysis technique for EV batteries to detect risk of fire or explosion
Recently, there have been a number of electric vehicle (EV) battery fire incidents. Unlike the batteries used in small mobile devices, such as smartphones, the battery pack of an EV is composed of hundreds of battery cells, and any instability can cause major casualties and property damage. Amid efforts to pinpoint the cause of battery fires, Korean researchers have developed a new analysis method to evaluate the thermal stability of EV batteries.
A research team at the Korea Institute of Science and Technology (KIST) led by Dr. Wonyoung Chang of the KIST Center for Energy Storage Research and Dr. Seung Min Kim of the Carbon Composite Materials Research Center of the Jeonbuk Branch built a real-time analysis platform for evaluating the thermal stability of the cathode material of a battery using a transmission electron microscope, through which they have identified the thermal degradation mechanism according to the changes in the chemical composition of the high-nickel cathode material for EVs.
The cathode material of an EV battery is one of the key factors that determine the battery capacity and thus the driving range. The cathode is manufactured by mixing various metals such as nickel, cobalt and aluminum or nickel, manganese, and cobalt according to the designated mixing ratio, and researchers from companies and academic circles are endeavoring to find an appropriate composition ratio to increase the driving range of EVs.
The cathode material contains nickel, and there is a positive correlation between the nickel content and the capacity. Also, since nickel is relatively cheaper than cobalt, adding more nickel lowers the unit cost of an EV battery. However, a fatal weakness of nickel is that it reduces the stability of the battery as it reacts easily to the external environment. In the case of the cathode material used for the third-generation EVs, which are currently in development, the nickel content has been increased to 80% or higher, and the consequent deterioration in stability must be improved to ensure safety.
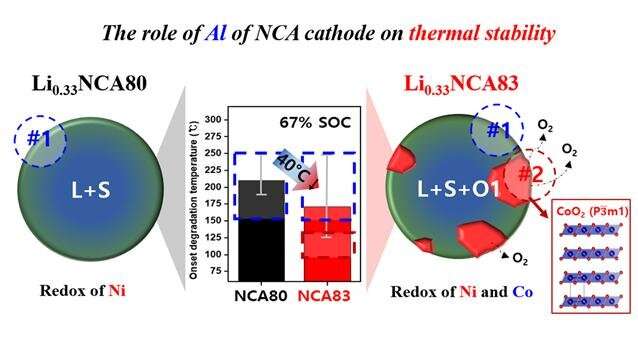
Battery fires are primarily initiated by a violent exothermic reaction between the charged oxide-based cathode material and the flammable liquid electrolytes. Thus, the research team at KIST focused on the surface of the cathode in contact with the electrolytes and applied a variety of transmission electron microscopy methods (electron energy loss spectroscopy, electron diffraction, etc.) for close observation and analysis of the crystal structure of the cathode and the chemical changes in the constituents resulting from a temperature increase. As a result, it was possible to identify the cause of reduced thermal stability of the battery based on the chemical composition of the NCA (nickel, cobalt, aluminum) cathode material and the role of the constituent elements to ensure the safety of the battery.
The KIST research team studied whether an increase in nickel compared to the aluminum content in the NCA cathode material improves the battery capacity, but found that it significantly reduces thermal stability in the upper charging limit (67% reaction of total lithium ions). An analysis showed that the lack of aluminum participating in the redox reaction led to the formation of a new phase (O1 phase) that can reduce thermal stability during the charging process. The surface of the new unstable phase was found to be the ultimate cause of reduced thermal stability.
Dr. Wonyoung Chang from KIST said, "There have been a series of EV fires worldwide, and the source of the fire, in many cases, was the battery. This study confirmed the importance of a chemical composition design that ensures thermal stability in developing high-performance cathode materials."
Dr. Seung Min Kim from the Jeonbuk Branch of KIST said, "Ensuring thermal stability of the cathode material where exothermic reactions begin is key to popularizing EVs. The advanced analysis techniques developed in this study will help determine the effects of elements in trace amounts and in turn lead to the development of high-performance cathode materials that are guaranteed to be safe."
More information: Eunmi Jo et al, Different thermal degradation mechanisms: Role of aluminum in Ni-rich layered cathode materials, Nano Energy (2020). DOI: 10.1016/j.nanoen.2020.105367