April 14, 2021 report
Membrane design prevents crossover in polysulfide redox flow batteries
Two researchers at the Chinese University of Hong Kong have developed a new membrane design that prevents crossover in polysulfide redox flow batteries. In their paper published in the journal Nature Energy, Zhejun Li and Yi-Chun Lu describe their work with the new membrane design. Wei Wang with Northwest National Laboratory has published a News & Views piece in the same journal issue describing problems with polysulfide redox flow batteries and the work by Li and Lu to overcome them.
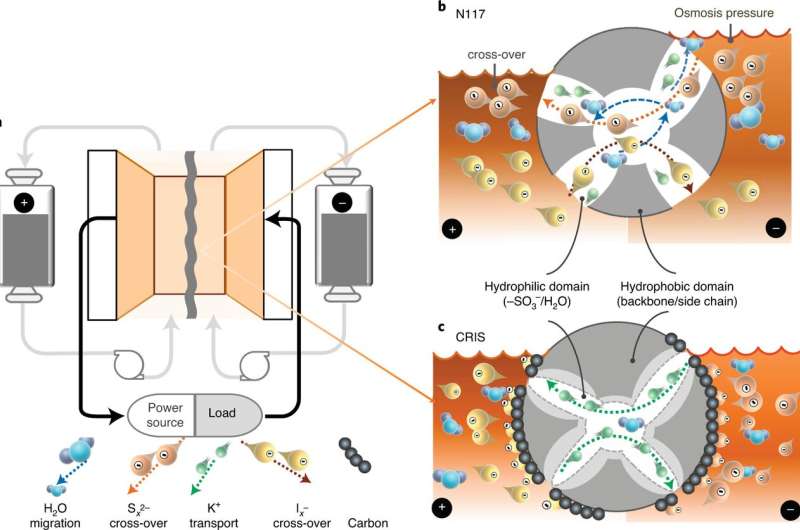
Back in the mid-1970s, NASA engineer Lawrence Thaller invented the idea of a redox flow battery. Since that time, such batteries have been widely used in large-scale applications such as backing up power grids. This is because they are easy to scale up to huge levels—the concept involves storing electrolytes in decentralized external tanks. In more recent times, scientists have wanted to build polysulfide redox flow batteries because they would be cheaper to build and maintain. A plant was built to test the idea, but unfortunately, doing so revealed a glaring weakness of the material—crossover of polysulfide species, which led to a very short life cycle. In this new effort, the researchers have overcome the problem of crossover with the development of a new membrane design.
The design involves using a layer of Ketjen black carbon affixed to the surface of a Nafion membrane using polyvinylidene fluoride. The researchers note this was done by drop casting the carbon slurries onto both sides of the membrane. They also note that because the membrane was so large, the surface area across it was able to serve as a trap for polyiodide and polysulfide ions. When saturation occurred, they pointed out, the anions were blocked, keeping them from moving onto the membrane, and thus no crossover could occur. The researchers note that the high conductivity of the carbon on the membrane overcomes the low conductivity of the underlying membrane; thus, it is not detrimental to the function of the membrane as would be the case in membranes where trapping typically occurs.
The researchers tested the design in both flow and static polysulfide–iodide cells and found that crossover did not occur, which meant improved life cycle numbers—proving the design feasible.
More information: Zhejun Li et al. Polysulfide-based redox flow batteries with long life and low levelized cost enabled by charge-reinforced ion-selective membranes, Nature Energy (2021). DOI: 10.1038/s41560-021-00804-x
Wei Wang. A membrane with repelling power, Nature Energy (2021). DOI: 10.1038/s41560-021-00811-y
© 2021 Science X Network